Inhibition of the Expression of the Small Heat Shock Protein αB-Crystallin Inhibits Exosome Secretion in Human Retinal Pigment Epithelial Cells in Culture
- PMID: 27129211
- PMCID: PMC4933212
- DOI: 10.1074/jbc.M115.698530
Inhibition of the Expression of the Small Heat Shock Protein αB-Crystallin Inhibits Exosome Secretion in Human Retinal Pigment Epithelial Cells in Culture
Abstract
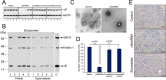
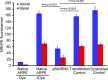
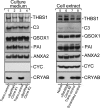
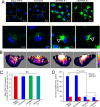
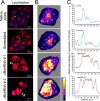
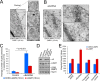
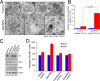
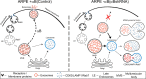
Exosomes carry cell type-specific molecular cargo to extracellular destinations and therefore act as lateral vectors of intercellular communication and transfer of genetic information from one cell to the other. We have shown previously that the small heat shock protein αB-crystallin (αB) is exported out of the adult human retinal pigment epithelial cells (ARPE19) packaged in exosomes. Here, we demonstrate that inhibition of the expression of αB via shRNA inhibits exosome secretion from ARPE19 cells indicating that exosomal cargo may have a role in exosome biogenesis (synthesis and/or secretion). Sucrose density gradient fractionation of the culture medium and cellular extracts suggests continued synthesis of exosomes but an inhibition of exosome secretion. In cells where αB expression was inhibited, the distribution of CD63 (LAMP3), an exosome marker, is markedly altered from the normal dispersed pattern to a stacked perinuclear presence. Interestingly, the total anti-CD63(LAMP3) immunofluorescence in the native and αB-inhibited cells remains unchanged suggesting continued exosome synthesis under conditions of impaired exosome secretion. Importantly, inhibition of the expression of αB results in a phenotype of the RPE cell that contains an increased number of vacuoles and enlarged (fused) vesicles that show increased presence of CD63(LAMP3) and LAMP1 indicating enhancement of the endolysosomal compartment. This is further corroborated by increased Rab7 labeling of this compartment (RabGTPase 7 is known to be associated with late endosome maturation). These data collectively point to a regulatory role for αB in exosome biogenesis possibly via its involvement at a branch point in the endocytic pathway that facilitates secretion of exosomes.
Keywords: CD63; LAMP1; Rab5; Rab7; endosome; exosome complex; lysosome; retinal pigment epithelium; secretion; small heat shock protein (sHsp).
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures


Similar articles
-
Mutant Huntingtin Inhibits αB-Crystallin Expression and Impairs Exosome Secretion from Astrocytes.J Neurosci. 2017 Sep 27;37(39):9550-9563. doi: 10.1523/JNEUROSCI.1418-17.2017. Epub 2017 Sep 11. J Neurosci. 2017. PMID: 28893927 Free PMC article.
-
AlphaB-crystallin is found in detergent-resistant membrane microdomains and is secreted via exosomes from human retinal pigment epithelial cells.J Biol Chem. 2011 Feb 4;286(5):3261-9. doi: 10.1074/jbc.M110.160135. Epub 2010 Nov 19. J Biol Chem. 2011. PMID: 21097504 Free PMC article.
-
αB crystallin is apically secreted within exosomes by polarized human retinal pigment epithelium and provides neuroprotection to adjacent cells.PLoS One. 2010 Oct 8;5(10):e12578. doi: 10.1371/journal.pone.0012578. PLoS One. 2010. PMID: 20949024 Free PMC article.
-
Alpha crystallins in the retinal pigment epithelium and implications for the pathogenesis and treatment of age-related macular degeneration.Biochim Biophys Acta. 2016 Jan;1860(1 Pt B):258-68. doi: 10.1016/j.bbagen.2015.05.016. Epub 2015 May 27. Biochim Biophys Acta. 2016. PMID: 26026469 Free PMC article. Review.
-
Controlled exosome release from the retinal pigment epithelium in situ.Exp Eye Res. 2014 Dec;129:1-4. doi: 10.1016/j.exer.2014.10.010. Epub 2014 Oct 11. Exp Eye Res. 2014. PMID: 25311167 Review.
Cited by
-
The Emerging Role of Neural Cell-Derived Exosomes in Intercellular Communication in Health and Neurodegenerative Diseases.Front Neurosci. 2021 Aug 31;15:738442. doi: 10.3389/fnins.2021.738442. eCollection 2021. Front Neurosci. 2021. PMID: 34531720 Free PMC article. Review.
-
Accumulation of cholesterol and increased demand for zinc in serum-deprived RPE cells.Mol Vis. 2016 Dec 10;22:1387-1404. eCollection 2016. Mol Vis. 2016. PMID: 28003730 Free PMC article.
-
The small heat shock proteins, HSPB1 and HSPB5, interact differently with lipid membranes.Cell Stress Chaperones. 2019 Sep;24(5):947-956. doi: 10.1007/s12192-019-01021-y. Epub 2019 Jul 23. Cell Stress Chaperones. 2019. PMID: 31338686 Free PMC article.
-
Extracellular small heat shock proteins: exosomal biogenesis and function.Cell Stress Chaperones. 2018 May;23(3):441-454. doi: 10.1007/s12192-017-0856-z. Epub 2017 Oct 30. Cell Stress Chaperones. 2018. PMID: 29086335 Free PMC article. Review.
-
Mutant Huntingtin Inhibits αB-Crystallin Expression and Impairs Exosome Secretion from Astrocytes.J Neurosci. 2017 Sep 27;37(39):9550-9563. doi: 10.1523/JNEUROSCI.1418-17.2017. Epub 2017 Sep 11. J Neurosci. 2017. PMID: 28893927 Free PMC article.
References
-
- Hanson P. I., and Cashikar A. (2012) Multivesicular body morphogenesis. Annu. Rev. Cell Dev. Biol. 28, 337–362 - PubMed
-
- Simons M., and Raposo G. (2009) Exosomes–vesicular carriers for intercellular communication. Curr. Opin. Cell Biol. 21, 575–581 - PubMed
-
- Zhang M., and Schekman R. (2013) Cell biology. Unconventional secretion, unconventional solutions. Science 340, 559–561 - PubMed
-
- Bok D. (1985) Retinal photoreceptor-pigment epithelium interactions. Friedenwald lecture. Invest. Ophthalmol. Vis. Sci. 26, 1659–1694 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

