Blue-Light Inhibition of Listeria monocytogenes Growth Is Mediated by Reactive Oxygen Species and Is Influenced by σB and the Blue-Light Sensor Lmo0799
- PMID: 27129969
- PMCID: PMC4907204
- DOI: 10.1128/AEM.00685-16
Blue-Light Inhibition of Listeria monocytogenes Growth Is Mediated by Reactive Oxygen Species and Is Influenced by σB and the Blue-Light Sensor Lmo0799
Abstract
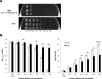
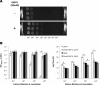
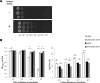
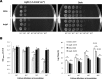
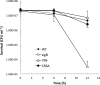
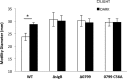
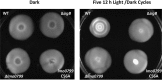
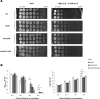
Listeria monocytogenes senses blue light via the flavin mononucleotide-containing sensory protein Lmo0799, leading to activation of the general stress response sigma factor SigB (σ(B)). In this study, we investigated the physiological response of this foodborne pathogen to blue light. We show that blue light (460 to 470 nm) doses of 1.5 to 2 mW cm(-2) cause inhibition of growth on agar-based and liquid culture media. The inhibitory effects are dependent on cell density, with reduced effects evident when high cell numbers are present. The addition of 20 mM dimethylthiourea, a scavenger of reactive oxygen species, or catalase to the medium reverses the inhibitory effects of blue light, suggesting that growth inhibition is mediated by the formation of reactive oxygen species. A mutant strain lacking σ(B) (ΔsigB) was found to be less inhibited by blue light than the wild type, likely indicating the energetic cost of deploying the general stress response. When a lethal dose of light (8 mW cm(-2)) was applied to cells, the ΔsigB mutant displayed a marked increase in sensitivity to light compared to the wild type. To investigate the role of the blue-light sensor Lmo0799, mutants were constructed that either had a deletion of the gene (Δlmo0799) or alteration in a conserved cysteine residue at position 56, which is predicted to play a pivotal role in the photocycle of the protein (lmo0799 C56A). Both mutants displayed phenotypes similar to the ΔsigB mutant in the presence of blue light, providing genetic evidence that residue 56 is critical for light sensing in L. monocytogenes Taken together, these results demonstrate that L. monocytogenes is inhibited by blue light in a manner that depends on reactive oxygen species, and they demonstrate clear light-dependent phenotypes associated with σ(B) and the blue-light sensor Lmo0799.
Importance: Listeria monocytogenes is a bacterial foodborne pathogen that can cause life-threatening infections in humans. It is known to be able to sense and respond to visible light. In this study, we examine the effects of blue light on the growth and survival of this pathogen. We show that growth can be inhibited at comparatively low doses of blue light, and that at higher doses, L. monocytogenes cells are killed. We present evidence suggesting that blue light inhibits this organism by causing the production of reactive oxygen species, such as hydrogen peroxide. We help clarify the mechanism of light sensing by constructing a "blind" version of the blue-light sensor protein. Finally, we show that activation of the general stress response by light has a negative effect on growth, probably because cellular resources are diverted into protective mechanisms rather than growth.
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures

Similar articles
-
Blue and red light modulates SigB-dependent gene transcription, swimming motility and invasiveness in Listeria monocytogenes.PLoS One. 2011 Jan 11;6(1):e16151. doi: 10.1371/journal.pone.0016151. PLoS One. 2011. PMID: 21264304 Free PMC article.
-
Mild Stress Conditions during Laboratory Culture Promote the Proliferation of Mutations That Negatively Affect Sigma B Activity in Listeria monocytogenes.J Bacteriol. 2020 Apr 9;202(9):e00751-19. doi: 10.1128/JB.00751-19. Print 2020 Apr 9. J Bacteriol. 2020. PMID: 32094160 Free PMC article.
-
Cycles of light and dark co-ordinate reversible colony differentiation in Listeria monocytogenes.Mol Microbiol. 2013 Feb;87(4):909-24. doi: 10.1111/mmi.12140. Epub 2013 Jan 21. Mol Microbiol. 2013. PMID: 23331346 Free PMC article.
-
Role and regulation of the stress activated sigma factor sigma B (σB) in the saprophytic and host-associated life stages of Listeria monocytogenes.Adv Appl Microbiol. 2019;106:1-48. doi: 10.1016/bs.aambs.2018.11.001. Epub 2018 Dec 24. Adv Appl Microbiol. 2019. PMID: 30798801 Review.
-
Identification and analysis of the osmotolerance associated genes in Listeria monocytogenes.Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 2008 Sep;25(9):1089-94. doi: 10.1080/02652030802056634. Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 2008. PMID: 19238619 Review.
Cited by
-
Genetic Factors Affect the Survival and Behaviors of Selected Bacteria during Antimicrobial Blue Light Treatment.Int J Mol Sci. 2021 Sep 28;22(19):10452. doi: 10.3390/ijms221910452. Int J Mol Sci. 2021. PMID: 34638788 Free PMC article. Review.
-
The Role of Stress and Stress Adaptations in Determining the Fate of the Bacterial Pathogen Listeria monocytogenes in the Food Chain.Front Microbiol. 2016 Nov 23;7:1865. doi: 10.3389/fmicb.2016.01865. eCollection 2016. Front Microbiol. 2016. PMID: 27933042 Free PMC article. Review.
-
The Effect of Atmospheric Cold Plasma on Bacterial Stress Responses and Virulence Using Listeria monocytogenes Knockout Mutants.Front Microbiol. 2019 Dec 11;10:2841. doi: 10.3389/fmicb.2019.02841. eCollection 2019. Front Microbiol. 2019. PMID: 31921026 Free PMC article.
-
Blue Light Sensing in Listeria monocytogenes Is Temperature-Dependent and the Transcriptional Response to It Is Predominantly SigB-Dependent.Front Microbiol. 2019 Nov 14;10:2497. doi: 10.3389/fmicb.2019.02497. eCollection 2019. Front Microbiol. 2019. PMID: 31798538 Free PMC article.
-
Acid stress signals are integrated into the σB-dependent general stress response pathway via the stressosome in the food-borne pathogen Listeria monocytogenes.PLoS Pathog. 2022 Mar 11;18(3):e1010213. doi: 10.1371/journal.ppat.1010213. eCollection 2022 Mar. PLoS Pathog. 2022. PMID: 35275969 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

