A 52-week pilot study of the effects of exenatide on body weight in patients with hypothalamic obesity
- PMID: 27133664
- PMCID: PMC4882247
- DOI: 10.1002/oby.21493
A 52-week pilot study of the effects of exenatide on body weight in patients with hypothalamic obesity
Abstract
Objective: Hypothalamic obesity (HO) is a common complication of hypothalamic tumors, and effective therapies are lacking. The objective of this pilot study was to investigate changes in body weight before and during treatment with exenatide.
Methods: This was a prospective, open-label, 52-week pilot study of exenatide (10 mcg b.i.d.) in adults with HO. Ten patients enrolled, and eight completed the study. Study measures included indirect calorimetry, body composition, buffet meals, diet recall, actigraphy, and hormone assays.
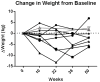
Results: Participants had obesity with a baseline weight of 137.2 ± 37.6 kg. Exenatide therapy was well tolerated. Change in weight with exenatide therapy was not significant (-1.4 ± 4.3 kg [95% CI -4.9 to 2.2], P = 0.40), but six out of eight completers lost weight (-6.2 to -0.2 kg). Participants reported significantly lower intake on food recall during treatment compared with baseline (7837.8 ± 2796.6 vs. 6258.4 ± 1970.7 kJ [95% CI -2915.8 to -242.6], P = 0.027), but there was no change in intake during buffet meals.
Conclusions: Significant weight loss was not observed in patients with HO treated with exenatide, but 75% of completers had stable or decreasing weight. Further studies are needed to evaluate weight loss efficacy in patients with HO.
Trial registration: ClinicalTrials.gov NCT01484873.
© 2016 The Obesity Society.
Figures
Similar articles
-
Antiobesity action of peripheral exenatide (exendin-4) in rodents: effects on food intake, body weight, metabolic status and side-effect measures.Int J Obes (Lond). 2006 Sep;30(9):1332-40. doi: 10.1038/sj.ijo.0803284. Epub 2006 Mar 14. Int J Obes (Lond). 2006. PMID: 16534527
-
Effects of a single dose of exenatide on appetite, gut hormones, and glucose homeostasis in adults with Prader-Willi syndrome.J Clin Endocrinol Metab. 2011 Aug;96(8):E1314-9. doi: 10.1210/jc.2011-0038. Epub 2011 Jun 1. J Clin Endocrinol Metab. 2011. PMID: 21632815 Clinical Trial.
-
Experiences with Glucagon-Like Peptide-1 Receptor Agonist in Children with Acquired Hypothalamic Obesity.Obes Facts. 2020;13(4):361-370. doi: 10.1159/000509302. Epub 2020 Aug 11. Obes Facts. 2020. PMID: 32781455 Free PMC article.
-
Exenatide as a novel weight loss modality in patients without diabetes.Ann Pharmacother. 2012 Dec;46(12):1700-6. doi: 10.1345/aph.1R372. Epub 2012 Nov 27. Ann Pharmacother. 2012. PMID: 23191935 Review.
-
Treatment of hypothalamic obesity in people with hypothalamic injury: new drugs are on the horizon.Front Endocrinol (Lausanne). 2023 Sep 13;14:1256514. doi: 10.3389/fendo.2023.1256514. eCollection 2023. Front Endocrinol (Lausanne). 2023. PMID: 37780616 Free PMC article. Review.
Cited by
-
Treatment of Hypothalamic Obesity With GLP-1 Analogs.J Endocr Soc. 2024 Nov 14;9(1):bvae200. doi: 10.1210/jendso/bvae200. eCollection 2024 Nov 26. J Endocr Soc. 2024. PMID: 39703362 Free PMC article. Review.
-
Semaglutide treatment in hypothalamic obesity: Two-Year outcomes on body composition, appetite, and quality of life.Pituitary. 2025 Aug 19;28(5):93. doi: 10.1007/s11102-025-01564-7. Pituitary. 2025. PMID: 40830718 Free PMC article.
-
A phase 3 randomized clinical trial using a once-weekly glucagon-like peptide-1 receptor agonist in adolescents and young adults with hypothalamic obesity.Diabetes Obes Metab. 2021 Feb;23(2):363-373. doi: 10.1111/dom.14224. Epub 2020 Oct 25. Diabetes Obes Metab. 2021. PMID: 33026160 Free PMC article. Clinical Trial.
-
Management of Acquired Hypothalamic Obesity After Childhood-Onset Craniopharyngioma-A Narrative Review.Biomedicines. 2025 Apr 22;13(5):1016. doi: 10.3390/biomedicines13051016. Biomedicines. 2025. PMID: 40426846 Free PMC article. Review.
-
Differential Role of Adipose Tissues in Obesity and Related Metabolic and Vascular Complications.Int J Endocrinol. 2016;2016:1216783. doi: 10.1155/2016/1216783. Epub 2016 Sep 27. Int J Endocrinol. 2016. PMID: 27766104 Free PMC article. Review.
References
-
- Bulow B, Attewell R, Hagmar L, Malmstrom P, Nordstrom CH, Erfurth EM. Postoperative prognosis in craniopharyngioma with respect to cardiovascular mortality, survival, and tumor recurrence. J Clin Endocrinol Metab. 1998;83(11):3897–904. Epub 1998/11/14. - PubMed
-
- Kim RJ, Shah R, Tershakovec AM, Zemel BS, Sutton LN, Grimberg A, et al. Energy expenditure in obesity associated with craniopharyngioma. Child’s nervous system: ChNS: official journal of the International Society for Pediatric Neurosurgery. 2010;26(7):913–7. doi: 10.1007/s00381-009-1078-1. Epub 2010/01/29. - DOI - PMC - PubMed
-
- Guran T, Turan S, Bereket A, Akcay T, Unluguzel G, Bas F, et al. The role of leptin, soluble leptin receptor, resistin, and insulin secretory dynamics in the pathogenesis of hypothalamic obesity in children. Eur J Pediatr. 2009;168(9):1043–8. doi: 10.1007/s00431-008-0876-x. Epub 2008/12/02. - DOI - PubMed
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical