Food-derived hydrophilic antioxidant ergothioneine is distributed to the brain and exerts antidepressant effect in mice
- PMID: 27134772
- PMCID: PMC4842932
- DOI: 10.1002/brb3.477
Food-derived hydrophilic antioxidant ergothioneine is distributed to the brain and exerts antidepressant effect in mice
Abstract
Background: Clinically used antidepressants suffer from various side effects. Therefore, we searched for a safe antidepressant with minimal side effects among food ingredients that are distributed to the brain. Here, we focused on ERGO (ergothioneine), which is a hydrophilic antioxidant and contained at high levels in edible golden oyster mushrooms. ERGO is a typical substrate of carnitine/organic cation transporter OCTN1/SLC22A4, which is expressed in the brain and neuronal stem cells, although little is known about its permeation through the BBB (blood-brain barrier) or its neurological activity.
Methods: To clarify the exposure of ERGO to brain and the possible antidepressant-like effect after oral ingestion, ERGO or GOME (golden oyster mushroom extract) which contains 1.2% (w/w) ERGO was mixed with feed and provided to mice for 2 weeks, and then ERGO concentration and antidepressant-like effect were evaluated by LC-MS/MS and FST (forced swimming test) or TST (tail suspension test), respectively.
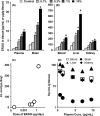
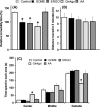
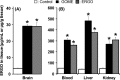
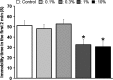
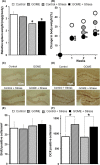
Results: Diet containing ERGO or GOME greatly increased the ERGO concentrations in plasma and brain, and significantly decreased the immobility time in both FST and TST. The required amount of GOME (~37 mg/day) to show the antidepressant-like effect corresponds to at most 8 g/day in humans. In mice receiving GOME-containing diet, doublecortin-positive cells showed a significant increase from the basal level, suggesting promotion of neuronal differentiation.
Conclusion: Thus, orally ingested ERGO is transported across the BBB into the brain, where it may promote neuronal differentiation and alleviate symptoms of depression at plausibly achieved level of daily ingestion.
Keywords: Antidepressant effect; OCTN1; depression; disposition; ergothioneine; neuronal differentiation.
Figures

Similar articles
-
Oral Administration of the Food-Derived Hydrophilic Antioxidant Ergothioneine Enhances Object Recognition Memory in Mice.Curr Mol Pharmacol. 2021;14(2):220-233. doi: 10.2174/1874467213666200212102710. Curr Mol Pharmacol. 2021. PMID: 32048982
-
Ergothioneine in the brain.FEBS Lett. 2022 May;596(10):1290-1298. doi: 10.1002/1873-3468.14271. Epub 2022 Jan 10. FEBS Lett. 2022. PMID: 34978075 Review.
-
Organic cation transporter Octn1-mediated uptake of food-derived antioxidant ergothioneine into infiltrating macrophages during intestinal inflammation in mice.Drug Metab Pharmacokinet. 2015 Jun;30(3):231-9. doi: 10.1016/j.dmpk.2015.02.003. Epub 2015 Feb 25. Drug Metab Pharmacokinet. 2015. PMID: 26003890
-
Ergothioneine and central nervous system diseases.Neurochem Res. 2022 Sep;47(9):2513-2521. doi: 10.1007/s11064-022-03665-2. Epub 2022 Jul 5. Neurochem Res. 2022. PMID: 35788879 Review.
-
Protective Effects of Pleurotus Species on UVB-Induced Skin Disorders at Clinically Relevant Plasma Concentrations of the Antioxidant Ergothioneine in Hairless Mice.Biol Pharm Bull. 2025;48(5):672-681. doi: 10.1248/bpb.b24-00893. Biol Pharm Bull. 2025. PMID: 40399103
Cited by
-
Systematic metabolic characterization of mental disorders reveals age-related metabolic disturbances as potential risk factors for depression in older adults.MedComm (2020). 2022 Sep 30;3(4):e165. doi: 10.1002/mco2.165. eCollection 2022 Dec. MedComm (2020). 2022. PMID: 36204590 Free PMC article.
-
Ergothioneine promotes longevity and healthy aging in male mice.Geroscience. 2024 Aug;46(4):3889-3909. doi: 10.1007/s11357-024-01111-5. Epub 2024 Mar 6. Geroscience. 2024. PMID: 38446314 Free PMC article.
-
A novel antioxidant ergothioneine PET radioligand for in vivo imaging applications.Sci Rep. 2021 Sep 16;11(1):18450. doi: 10.1038/s41598-021-97925-w. Sci Rep. 2021. PMID: 34531467 Free PMC article.
-
Ganoderma Fusions with High Yield of Ergothioneine and Comparative Analysis of Its Genomics.J Fungi (Basel). 2023 Nov 2;9(11):1072. doi: 10.3390/jof9111072. J Fungi (Basel). 2023. PMID: 37998877 Free PMC article.
-
Reactive oxygen species, toxicity, oxidative stress, and antioxidants: chronic diseases and aging.Arch Toxicol. 2023 Oct;97(10):2499-2574. doi: 10.1007/s00204-023-03562-9. Epub 2023 Aug 19. Arch Toxicol. 2023. PMID: 37597078 Free PMC article. Review.
References
-
- Alusik, S. , Kalatova D., and Paluch Z.. 2014. Serotonin syndrome. Neuro. Endocrinol. Lett. 35:265–273. - PubMed
-
- Aruoma, O. I. , Spencer J. P., and Mahmood N.. 1999. Protection against oxidative damage and cell death by the natural antioxidant ergothioneine. Food Chem. Toxicol. 37:1043–1053. - PubMed
-
- Bet, P. M. , Hugtenburg J. G., Penninx B. W., and Hoogendijk W. J.. 2013. Side effects of antidepressants during long‐term use in a naturalistic setting. Eur. Neuropsychopharmacol. 23:1443–1451. - PubMed
-
- Bukhari, I. A. , and Dar A.. 2013. Behavioral profile of Hypericum perforatum (St. John's Wort) extract. A comparison with standard antidepressants in animal models of depression. Eur. Rev. Med. Pharmacol. Sci. 17:1082–1089. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

