Comparative Functional Alanine Positional Scanning of the α-Melanocyte Stimulating Hormone and NDP-Melanocyte Stimulating Hormone Demonstrates Differential Structure-Activity Relationships at the Mouse Melanocortin Receptors
- PMID: 27135265
- PMCID: PMC5596636
- DOI: 10.1021/acschemneuro.6b00098
Comparative Functional Alanine Positional Scanning of the α-Melanocyte Stimulating Hormone and NDP-Melanocyte Stimulating Hormone Demonstrates Differential Structure-Activity Relationships at the Mouse Melanocortin Receptors
Abstract
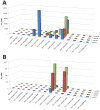
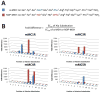
The melanocortin system has been implicated in the regulation of various physiological functions including melanogenesis, steroidogenesis, energy homeostasis, and feeding behavior. Five melanocortin receptors have been identified to date and belong to the family of G protein-coupled receptors (GPCR). Post-translational modification of the proopiomelanocortin (POMC) prohormone leads to the biosynthesis of the endogenous melanocortin agonists, including α-melanocyte stimulating hormone (α-MSH), β-MSH, γ-MSH, and adrenocorticotropic hormone (ACTH). All the melanocortin agonists derived from the POMC prohormone contain a His-Phe-Arg-Trp tetrapeptide sequence that has been implicated in eliciting the pharmacological responses at the melanocortin receptors. Herein, an alanine (Ala) positional scan is reported for the endogenous α-MSH ligand and the synthetic, more potent, NDP-MSH peptide (Ac-Ser(1)-Tyr(2)-Ser(3)-Nle(4)-Glu(5)-His(6)-DPhe(7)-Arg(8)-Trp(9)-Gly(10)-Lys(11)-Pro(12)-Val(13)-NH2) at the cloned mouse melanocortin receptors to test the assumption that the structure-activity relationships of one ligand would apply to the other. Several residues outside of the postulated pharmacophore altered potency at the melanocortin receptors, most notably the 1560-, 37-, and 15-fold potency loss when the Glu(5) position of α-MSH was substituted with Ala at the mMC1R, mMC3R, and mMC4R, respectively. Importantly, the altered potencies due to Ala substitutions in α-MSH did not necessarily correlate with equivalent Ala substitutions in NDP-MSH, indicating that structural modifications and corresponding biological activities in one of these melanocortin ligands may not be predictive for the other agonist.
Keywords: GPCR; Melanotropin; NDP-MSH; alanine positional scan; obesity; α-MSH.
Figures

Similar articles
-
Chimeric NDP-MSH and MTII melanocortin peptides with agouti-related protein (AGRP) Arg-Phe-Phe amino acids possess agonist melanocortin receptor activity.Peptides. 2003 Dec;24(12):1899-908. doi: 10.1016/j.peptides.2003.10.005. Peptides. 2003. PMID: 15127941
-
Biological and conformational examination of stereochemical modifications using the template melanotropin peptide, Ac-Nle-c[Asp-His-Phe-Arg-Trp-Ala-Lys]-NH2, on human melanocortin receptors.J Med Chem. 1997 May 23;40(11):1738-48. doi: 10.1021/jm960845e. J Med Chem. 1997. PMID: 9171884
-
Structure-activity relationships of the unique and potent agouti-related protein (AGRP)-melanocortin chimeric Tyr-c[beta-Asp-His-DPhe-Arg-Trp-Asn-Ala-Phe-Dpr]-Tyr-NH2 peptide template.J Med Chem. 2005 Apr 21;48(8):3060-75. doi: 10.1021/jm049010r. J Med Chem. 2005. PMID: 15828845
-
Melanocortins and cardiovascular regulation.Eur J Pharmacol. 1998 Oct 30;360(1):1-14. doi: 10.1016/s0014-2999(98)00615-3. Eur J Pharmacol. 1998. PMID: 9845266 Review.
-
111In-DOTA-Re(Cys3,4,10,d-Phe7,Arg11)αMSH3-13.2007 Sep 1 [updated 2007 Dec 17]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2007 Sep 1 [updated 2007 Dec 17]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 20641798 Free Books & Documents. Review.
Cited by
-
Bench-top to clinical therapies: A review of melanocortin ligands from 1954 to 2016.Biochim Biophys Acta Mol Basis Dis. 2017 Oct;1863(10 Pt A):2414-2435. doi: 10.1016/j.bbadis.2017.03.020. Epub 2017 Mar 29. Biochim Biophys Acta Mol Basis Dis. 2017. PMID: 28363699 Free PMC article. Review.
-
Discovery of Mixed Pharmacology Melanocortin-3 Agonists and Melanocortin-4 Receptor Tetrapeptide Antagonist Compounds (TACOs) Based on the Sequence Ac-Xaa1-Arg-(pI)DPhe-Xaa4-NH2.J Med Chem. 2017 May 25;60(10):4342-4357. doi: 10.1021/acs.jmedchem.7b00301. Epub 2017 May 15. J Med Chem. 2017. PMID: 28453292 Free PMC article.
-
The Parallel Structure-Activity Relationship Screening of Three Compounds Identifies the Common Agonist Pharmacophore of Pyrrolidine Bis-Cyclic Guanidine Melanocortin-3 Receptor (MC3R) Small-Molecule Ligands.Int J Mol Sci. 2023 Jun 14;24(12):10145. doi: 10.3390/ijms241210145. Int J Mol Sci. 2023. PMID: 37373293 Free PMC article.
-
Discovery of Nanomolar Melanocortin-3 Receptor (MC3R)-Selective Small Molecule Pyrrolidine Bis-Cyclic Guanidine Agonist Compounds Via a High-Throughput "Unbiased" Screening Campaign.J Med Chem. 2021 May 13;64(9):5577-5592. doi: 10.1021/acs.jmedchem.0c02041. Epub 2021 Apr 22. J Med Chem. 2021. PMID: 33886285 Free PMC article.
References
-
- Chhajlani V, Wikberg JE. Molecular cloning and expression of the human melanocyte stimulating hormone receptor cDNA. FEBS Lett. 1992;309:417–420. - PubMed
-
- Mountjoy KG, Robbins LS, Mortrud MT, Cone RD. The cloning of a family of genes that encode the melanocortin receptors. Science. 1992;257:1248–1251. - PubMed
-
- Gantz I, Miwa H, Konda Y, Shimoto Y, Tashiro T, Watson SJ, DelValle J, Yamada T. Molecular cloning, expression, and gene localization of a fourth melanocortin receptor. J Biol Chem. 1993;268:15174–15179. - PubMed
-
- Gantz I, Konda Y, Tashiro T, Shimoto Y, Miwa H, Munzert G, Watson SJ, DelValle J, Yamada T. Molecular cloning of a novel melanocortin receptor. J Biol Chem. 1993;268:8246–8250. - PubMed
-
- Mountjoy KG, Mortrud MT, Low MJ, Simerly RB, Cone RD. Localization of the melanocortin-4 receptor (MC4-R) in neuroendocrine and autonomic control circuits in the brain. Mol Endocrinol. 1994;8:1298–1308. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

