TPC2 controls pigmentation by regulating melanosome pH and size
- PMID: 27140606
- PMCID: PMC4878521
- DOI: 10.1073/pnas.1600108113
TPC2 controls pigmentation by regulating melanosome pH and size
Abstract
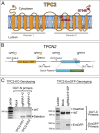
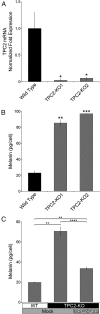
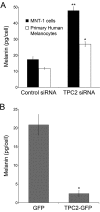
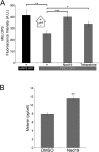
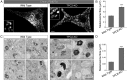
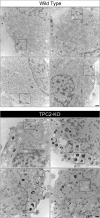
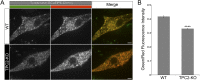
Melanin is responsible for pigmentation of skin and hair and is synthesized in a specialized organelle, the melanosome, in melanocytes. A genome-wide association study revealed that the two pore segment channel 2 (TPCN2) gene is strongly linked to pigmentation variations. TPCN2 encodes the two-pore channel 2 (TPC2) protein, a cation channel. Nevertheless, how TPC2 regulates pigmentation remains unknown. Here, we show that TPC2 is expressed in melanocytes and localizes to the melanosome-limiting membrane and, to a lesser extent, to endolysosomal compartments by confocal fluorescence and immunogold electron microscopy. Immunomagnetic isolation of TPC2-containing organelles confirmed its coresidence with melanosomal markers. TPCN2 knockout by means of clustered regularly interspaced short palindromic repeat/CRISPR-associated 9 gene editing elicited a dramatic increase in pigment content in MNT-1 melanocytic cells. This effect was rescued by transient expression of TPC2-GFP. Consistently, siRNA-mediated knockdown of TPC2 also caused a substantial increase in melanin content in both MNT-1 cells and primary human melanocytes. Using a newly developed genetically encoded pH sensor targeted to melanosomes, we determined that the melanosome lumen in TPC2-KO MNT-1 cells and primary melanocytes subjected to TPC2 knockdown is less acidic than in control cells. Fluorescence and electron microscopy analysis revealed that TPC2-KO MNT-1 cells have significantly larger melanosomes than control cells, but the number of organelles is unchanged. TPC2 likely regulates melanosomes pH and size by mediating Ca(2+) release from the organelle, which is decreased in TPC2-KO MNT-1 cells, as determined with the Ca(2+) sensor tyrosinase-GCaMP6. Thus, our data show that TPC2 regulates pigmentation through two fundamental determinants of melanosome function: pH and size.
Keywords: melanosome; membrane traffic; organelle pH; pigmentation; two-pore channel 2.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







Comment in
-
Ions switch off darkness: role of TPC2 in melanosomes.Pigment Cell Melanoma Res. 2016 Sep;29(5):498-9. doi: 10.1111/pcmr.12510. Pigment Cell Melanoma Res. 2016. PMID: 27412049 No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

