Evidence of In Vivo Existence of Borrelia Biofilm in Borrelial Lymphocytomas
- PMID: 27141311
- PMCID: PMC4838982
- DOI: 10.1556/1886.2015.00049
Evidence of In Vivo Existence of Borrelia Biofilm in Borrelial Lymphocytomas
Abstract
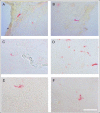
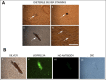
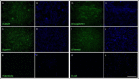
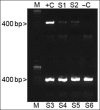
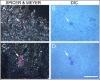
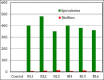
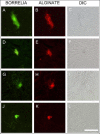
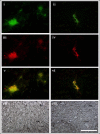
Lyme borreliosis, caused by the spirochete Borrelia burgdorferi sensu lato, has grown into a major public health problem. We recently identified a novel morphological form of B. burgdorferi, called biofilm, a structure that is well known to be highly resistant to antibiotics. However, there is no evidence of the existence of Borrelia biofilm in vivo; therefore, the main goal of this study was to determine the presence of Borrelia biofilm in infected human skin tissues. Archived skin biopsy tissues from borrelial lymphocytomas (BL) were reexamined for the presence of B. burgdorferi sensu lato using Borrelia-specific immunohistochemical staining (IHC), fluorescent in situ hybridization, combined fluorescent in situ hybridization (FISH)-IHC, polymerase chain reaction (PCR), and fluorescent and atomic force microscopy methods. Our morphological and histological analyses showed that significant amounts of Borrelia-positive spirochetes and aggregates exist in the BL tissues. Analyzing structures positive for Borrelia showed that aggregates, but not spirochetes, expressed biofilm markers such as protective layers of different mucopolysaccharides, especially alginate. Atomic force microscopy revealed additional hallmark biofilm features of the Borrelia/alginate-positive aggregates such as inside channels and surface protrusions. In summary, this is the first study that demonstrates the presence of Borrelia biofilm in human infected skin tissues.
Keywords: Lyme disease; alginate; atomic force microscopy; biofilm; mucopolysaccharides.
Conflict of interest statement
The authors have declared that no competing interest exists.
Figures



Similar articles
-
The Long-Term Persistence of Borrelia burgdorferi Antigens and DNA in the Tissues of a Patient with Lyme Disease.Antibiotics (Basel). 2019 Oct 11;8(4):183. doi: 10.3390/antibiotics8040183. Antibiotics (Basel). 2019. PMID: 31614557 Free PMC article.
-
Polymerase chain reaction in diagnosis of Borrelia burgdorferi infections and studies on taxonomic classification.APMIS Suppl. 2002;(105):1-40. APMIS Suppl. 2002. PMID: 11985118
-
Borrelia and Chlamydia Can Form Mixed Biofilms in Infected Human Skin Tissues.Eur J Microbiol Immunol (Bp). 2019 Apr 11;9(2):46-55. doi: 10.1556/1886.2019.00003. eCollection 2019 Jun 3. Eur J Microbiol Immunol (Bp). 2019. PMID: 31223496 Free PMC article.
-
[Lyme borreliosis--diagnostic difficulties in interpreting serological results].Med Pregl. 2010 Nov-Dec;63(11-12):839-43. doi: 10.2298/mpns1012839g. Med Pregl. 2010. Retraction in: Med Pregl. 2012 Nov-Dec;65(11-12):531. PMID: 21553464 Retracted. Review. Serbian.
-
Biological aspects of Lyme disease spirochetes: unique bacteria of the Borrelia burgdorferi species group.Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2007 Dec;151(2):175-86. doi: 10.5507/bp.2007.032. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2007. PMID: 18345249 Review.
Cited by
-
The Brilliance of Borrelia: Mechanisms of Host Immune Evasion by Lyme Disease-Causing Spirochetes.Pathogens. 2021 Mar 2;10(3):281. doi: 10.3390/pathogens10030281. Pathogens. 2021. PMID: 33801255 Free PMC article. Review.
-
Inhibition of Borrelia Burgdorferi-Induced TLR2-NFκB Canonical Signaling by Gallic Acid through Targeting the CD14+ Adaptor Protein and p65 Molecule.Int J Mol Sci. 2022 Sep 20;23(19):10987. doi: 10.3390/ijms231910987. Int J Mol Sci. 2022. PMID: 36232290 Free PMC article.
-
The Long-Term Persistence of Borrelia burgdorferi Antigens and DNA in the Tissues of a Patient with Lyme Disease.Antibiotics (Basel). 2019 Oct 11;8(4):183. doi: 10.3390/antibiotics8040183. Antibiotics (Basel). 2019. PMID: 31614557 Free PMC article.
-
Borrelia burgdorferi Co-Localizing with Amyloid Markers in Alzheimer's Disease Brain Tissues.J Alzheimers Dis. 2022;85(2):889-903. doi: 10.3233/JAD-215398. J Alzheimers Dis. 2022. PMID: 34897095 Free PMC article.
-
Screening Marine Microbial Metabolites as Promising Inhibitors of Borrelia garinii: A Structural Docking Approach towards Developing Novel Lyme Disease Treatment.Biomed Res Int. 2024 Feb 29;2024:9997082. doi: 10.1155/2024/9997082. eCollection 2024. Biomed Res Int. 2024. PMID: 38456098 Free PMC article.
References
-
- Mead PS: Epidemiology of Lyme Disease. Infect Dis Clin North Am 29, 187–210 (2015) - PubMed
-
- Liegner KB, Shapiro JR, Ramsay D, Halperin AJ, Hogrefe W, Kong L: Recurrent erythema migrans despite extended antibiotic treatment with minocycline in a patient with persisting Borrelia burgdorferi infection. J Am Acad Dermatol 28, 312–314 (1993) - PubMed
-
- Dumler JS: Molecular diagnosis of Lyme disease: review and meta-analysis. Mol Diagn 6, 1–11 (2001) - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
