Dorsal root ganglia in Friedreich ataxia: satellite cell proliferation and inflammation
- PMID: 27142428
- PMCID: PMC4855486
- DOI: 10.1186/s40478-016-0288-5
Dorsal root ganglia in Friedreich ataxia: satellite cell proliferation and inflammation
Abstract
Introduction: Dorsal root ganglia (DRG) are highly vulnerable to frataxin deficiency in Friedreich ataxia (FA), an autosomal recessive disease due to pathogenic homozygous guanine-adenine-adenine trinucleotide repeat expansions in intron 1 of the FXN gene (chromosome 9q21.11). An immunohistochemical and immunofluorescence study of DRG in 15 FA cases and 12 controls revealed that FA causes major primary changes in satellite cells and inflammatory destruction of neurons. A panel of antibodies was used to reveal the cytoplasm of satellite cells (glutamine synthetase, S100, metabotropic glutamate receptors 2/3, excitatory amino acid transporter 1, ATP-sensitive inward rectifier potassium channel 10, and cytosolic ferritin), gap junctions (connexin 43), basement membranes (laminin), mitochondria (ATP synthase subunit beta and frataxin), and monocytes (CD68 and IBA1).
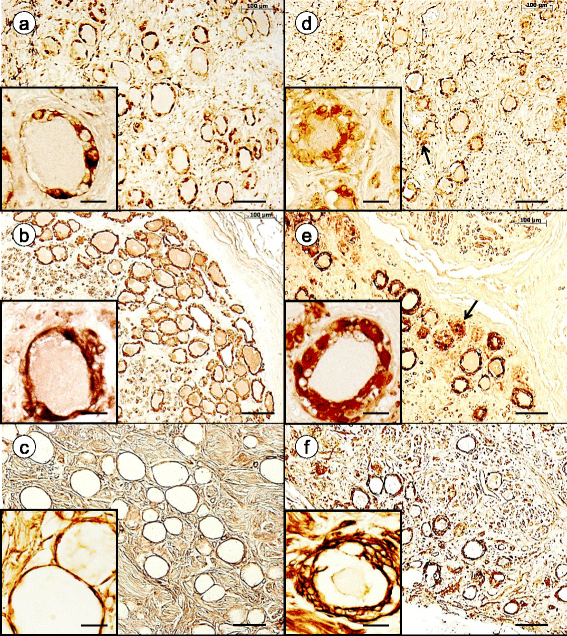
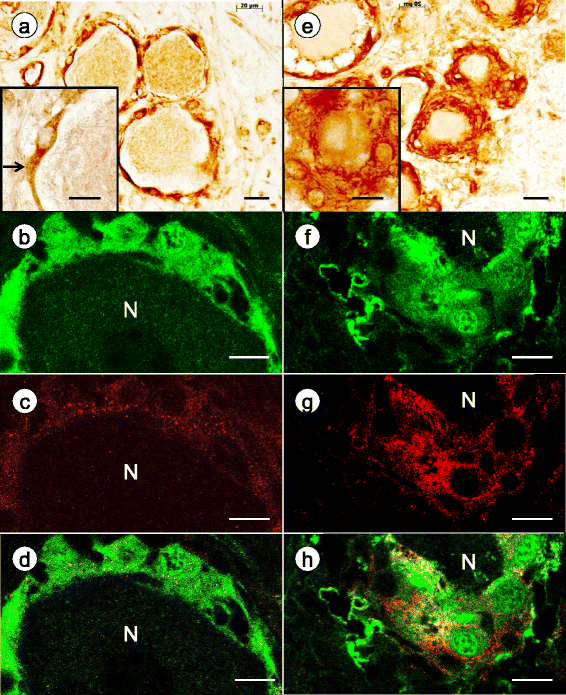
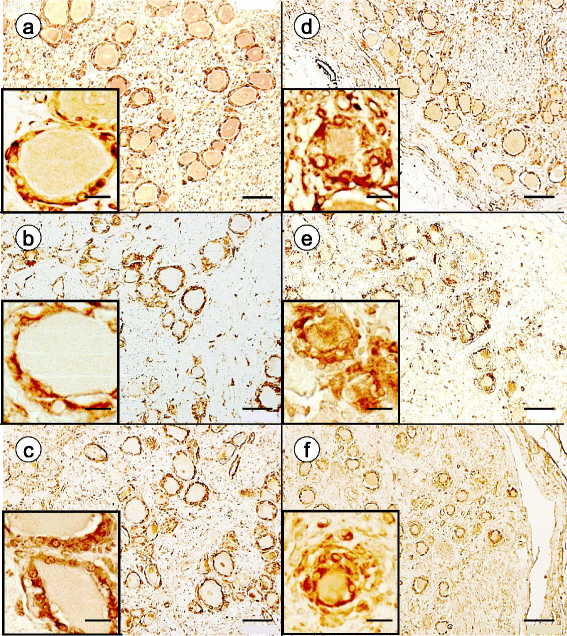
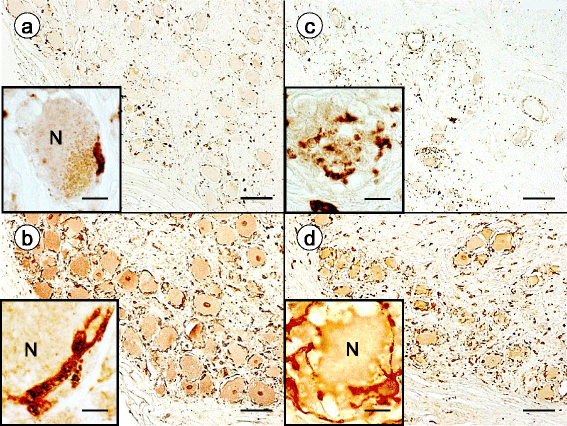
Results: Reaction product of the cytoplasmic markers and laminin confirmed proliferation of satellite cells and processes into multiple perineuronal layers and residual nodules. The formation of connexin 43-reactive gap junctions between satellite cells was strongly upregulated. Proliferating satellite cells in FA displayed many more frataxin- and ATP5B-reactive mitochondria than normal. Monocytes entered into the satellite cell layer, appeared to penetrate neuronal plasma membranes, and infiltrated residual nodules. Satellite cells and IBA1-reactive monocytes displayed upregulated ferritin biosynthesis, which was most likely due to leakage of iron from dying neurons.
Conclusions: We conclude that FA differentially affects the key cellular elements of DRG, and postulate that the disease causes loss of bidirectional trophic support between satellite cells and neurons.
Keywords: Dorsal root ganglion; Friedreich ataxia; Gap junction; Inflammation; Monocyte; Satellite cell.
Figures


Similar articles
-
Friedreich ataxia: metal dysmetabolism in dorsal root ganglia.Acta Neuropathol Commun. 2013 Jun 19;1:26. doi: 10.1186/2051-5960-1-26. Acta Neuropathol Commun. 2013. PMID: 24252376 Free PMC article.
-
The dorsal root ganglion in Friedreich's ataxia.Acta Neuropathol. 2009 Dec;118(6):763-76. doi: 10.1007/s00401-009-0589-x. Epub 2009 Aug 30. Acta Neuropathol. 2009. PMID: 19727777
-
Frataxin-deficient neurons and mice models of Friedreich ataxia are improved by TAT-MTScs-FXN treatment.J Cell Mol Med. 2018 Feb;22(2):834-848. doi: 10.1111/jcmm.13365. Epub 2017 Oct 5. J Cell Mol Med. 2018. PMID: 28980774 Free PMC article.
-
Friedreich ataxia: neuropathology revised.J Neuropathol Exp Neurol. 2013 Feb;72(2):78-90. doi: 10.1097/NEN.0b013e31827e5762. J Neuropathol Exp Neurol. 2013. PMID: 23334592 Free PMC article. Review.
-
Beyond loss of frataxin: the complex molecular pathology of Friedreich ataxia.Discov Med. 2014 Jan;17(91):25-35. Discov Med. 2014. PMID: 24411698 Review.
Cited by
-
The immune system as a driver of mitochondrial disease pathogenesis: a review of evidence.Orphanet J Rare Dis. 2022 Sep 2;17(1):335. doi: 10.1186/s13023-022-02495-3. Orphanet J Rare Dis. 2022. PMID: 36056365 Free PMC article. Review.
-
Redox homeostasis and inflammation in fibroblasts of patients with Friedreich Ataxia: a possible cross talk.Front Mol Neurosci. 2025 Apr 16;18:1571402. doi: 10.3389/fnmol.2025.1571402. eCollection 2025. Front Mol Neurosci. 2025. PMID: 40308559 Free PMC article.
-
An Intimate Role for Adult Dorsal Root Ganglia Resident Cycling Cells in the Generation of Local Macrophages and Satellite Glial Cells.J Neuropathol Exp Neurol. 2018 Oct 1;77(10):929-941. doi: 10.1093/jnen/nly072. J Neuropathol Exp Neurol. 2018. PMID: 30169768 Free PMC article.
-
Heart and Nervous System Pathology in Compound Heterozygous Friedreich Ataxia.J Neuropathol Exp Neurol. 2017 Aug 1;76(8):665-675. doi: 10.1093/jnen/nlx047. J Neuropathol Exp Neurol. 2017. PMID: 28789479 Free PMC article.
-
Central Nervous System Therapeutic Targets in Friedreich Ataxia.Hum Gene Ther. 2020 Dec;31(23-24):1226-1236. doi: 10.1089/hum.2020.264. Hum Gene Ther. 2020. PMID: 33238751 Free PMC article. Review.
References
-
- Albuerne M, Mammola CL, Naves FJ, Levanti B, Germanà G, Vega JA. Immunohistochemical localization of S100 proteins in dorsal root, sympathetic and enteric ganglia of several mammalian species, including man. J Peripher Nerv Syst. 1998;3:243–53. - PubMed
-
- Brumovsky P. VGLUTs in peripheral neurons and the spinal cord: time for a review. ISRN Neurol. 2013. http://dx.doi.org/10.1155/2013/829753. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

