Nuclear receptor REV-ERBα mediates circadian sensitivity to mortality in murine vesicular stomatitis virus-induced encephalitis
- PMID: 27143721
- PMCID: PMC4878505
- DOI: 10.1073/pnas.1520489113
Nuclear receptor REV-ERBα mediates circadian sensitivity to mortality in murine vesicular stomatitis virus-induced encephalitis
Abstract
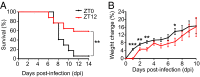
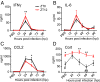
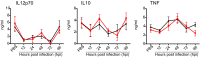
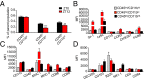
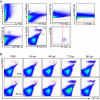
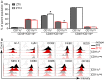
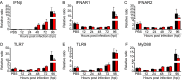
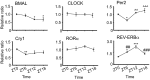
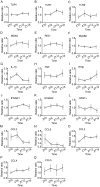
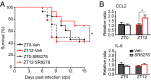
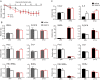
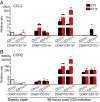
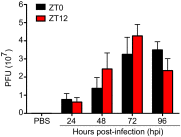
Certain components and functions of the immune system, most notably cytokine production and immune cell migration, are under circadian regulation. Such regulation suggests that circadian rhythms may have an effect on disease onset, progression, and resolution. In the vesicular stomatitis virus (VSV)-induced encephalitis model, the replication, caudal penetration, and survivability of intranasally applied VSV depends on both innate and adaptive immune mechanisms. In the current study, we investigated the effect of circadian time of infection on the progression and outcome of VSV-induced encephalitis and demonstrated a significant decrease in the survival rate in mice infected at the start of the rest cycle, zeitgeber time 0 (ZT0). The lower survival rate in these mice was associated with higher levels of circulating chemokine (C-C motif) ligand 2 (CCL2), a greater number of peripherally derived immune cells accumulating in the olfactory bulb (OB), and increased production of proinflammatory cytokines, indicating an immune-mediated pathology. We also found that the acrophase of molecular circadian clock component REV-ERBα mRNA expression in the OB coincides with the start of the active cycle, ZT12, when VSV infection results in a more favorable outcome. This result led us to hypothesize that REV-ERBα may mediate the circadian effect on survival following VSV infection. Blocking REV-ERBα activity before VSV administration resulted in a significant increase in the expression of CCL2 and decreased survival in mice infected at the start of the active cycle. These data demonstrate that REV-ERBα-mediated inhibition of CCL2 expression during viral-induced encephalitis may have a protective effect.
Keywords: circadian; immune response; inflammation; monocytes; vesicular stomatitis virus.
Figures
Similar articles
-
REV-ERBα and the clock gene machinery in mouse peripheral tissues: a possible role as a synchronizing hinge.J Biol Regul Homeost Agents. 2012 Apr-Jun;26(2):265-76. J Biol Regul Homeost Agents. 2012. PMID: 22824754
-
The nuclear receptor REV-ERBα mediates circadian regulation of innate immunity through selective regulation of inflammatory cytokines.Proc Natl Acad Sci U S A. 2012 Jan 10;109(2):582-7. doi: 10.1073/pnas.1106750109. Epub 2011 Dec 19. Proc Natl Acad Sci U S A. 2012. PMID: 22184247 Free PMC article.
-
A circadian clock gene, Rev-erbα, modulates the inflammatory function of macrophages through the negative regulation of Ccl2 expression.J Immunol. 2014 Jan 1;192(1):407-17. doi: 10.4049/jimmunol.1301982. Epub 2013 Dec 4. J Immunol. 2014. PMID: 24307731
-
Circadian clock proteins and immunity.Immunity. 2014 Feb 20;40(2):178-86. doi: 10.1016/j.immuni.2014.02.002. Immunity. 2014. PMID: 24560196 Review.
-
Rev-erbα and the circadian transcriptional regulation of metabolism.Diabetes Obes Metab. 2015 Sep;17 Suppl 1(0 1):12-6. doi: 10.1111/dom.12510. Diabetes Obes Metab. 2015. PMID: 26332963 Free PMC article. Review.
Cited by
-
The circadian clock influences T cell responses to vaccination by regulating dendritic cell antigen processing.Nat Commun. 2022 Dec 5;13(1):7217. doi: 10.1038/s41467-022-34897-z. Nat Commun. 2022. PMID: 36470865 Free PMC article.
-
Circadian rhythms in infectious diseases and symbiosis.Semin Cell Dev Biol. 2022 Jun;126:37-44. doi: 10.1016/j.semcdb.2021.09.004. Epub 2021 Oct 6. Semin Cell Dev Biol. 2022. PMID: 34625370 Free PMC article. Review.
-
REV-ERBα and REV-ERBβ function as key factors regulating Mammalian Circadian Output.Sci Rep. 2019 Jul 15;9(1):10171. doi: 10.1038/s41598-019-46656-0. Sci Rep. 2019. PMID: 31308426 Free PMC article.
-
The evolutionary ecology of circadian rhythms in infection.Nat Ecol Evol. 2019 Apr;3(4):552-560. doi: 10.1038/s41559-019-0831-4. Epub 2019 Mar 18. Nat Ecol Evol. 2019. PMID: 30886375 Free PMC article.
-
Circadian metabolic adaptations to infections.Philos Trans R Soc Lond B Biol Sci. 2025 Jan 23;380(1918):20230473. doi: 10.1098/rstb.2023.0473. Epub 2025 Jan 23. Philos Trans R Soc Lond B Biol Sci. 2025. PMID: 39842481 Review.
References
-
- Curtis AM, Bellet MM, Sassone-Corsi P, O’Neill LAJ. Circadian clock proteins and immunity. Immunity. 2014;40(2):178–186. - PubMed
-
- Sato S, et al. A circadian clock gene, Rev-erbα, modulates the inflammatory function of macrophages through the negative regulation of Ccl2 expression. J Immunol. 2014;192(1):407–417. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

