Veratri Nigri Rhizoma et Radix (Veratrum nigrum L.) and Its Constituent Jervine Prevent Adipogenesis via Activation of the LKB1-AMPKα-ACC Axis In Vivo and In Vitro
- PMID: 27143989
- PMCID: PMC4837256
- DOI: 10.1155/2016/8674397
Veratri Nigri Rhizoma et Radix (Veratrum nigrum L.) and Its Constituent Jervine Prevent Adipogenesis via Activation of the LKB1-AMPKα-ACC Axis In Vivo and In Vitro
Abstract
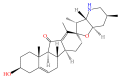
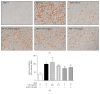
This study was performed in order to investigate the antiobese effects of the ethanolic extract of Veratri Nigri Rhizoma et Radix (VN), a herb with limited usage, due to its toxicology. An HPLC analysis identified jervine as a constituent of VN. By an Oil Red O assay and a Real-Time RT-PCR assay, VN showed higher antiadipogenic effects than jervine. In high-fat diet- (HFD-) induced obese C57BL/6J mice, VN administration suppressed body weight gain. The levels of peroxisome proliferator-activated receptor gamma (PPARγ), CCAAT-enhancer-binding protein alpha (C/EBPα), adipocyte fatty-acid-binding protein (aP2), adiponectin, resistin, and LIPIN1 were suppressed by VN, while SIRT1 was upregulated. Furthermore, VN activated phosphorylation of the liver kinase B1- (LKB1-) AMP-activated protein kinase alpha- (AMPKα-) acetyl CoA carboxylase (ACC) axis. Further investigation of cotreatment of VN with the AMPK agonist AICAR or AMPK inhibitor Compound C showed that VN can activate the phosphorylation of AMPKα in compensation to the inhibition of Compound C. In conclusion, VN shows antiobesity effects in HFD-induced obese C57BL/6J mice. In 3T3-L1 adipocytes, VN has antiadipogenic features, which is due to activating the LKB1-AMPKα-ACC axis. These results suggest that VN has a potential benefit in preventing obesity.
Figures




Similar articles
-
Moringa oleifera Leaf Petroleum Ether Extract Inhibits Lipogenesis by Activating the AMPK Signaling Pathway.Front Pharmacol. 2018 Dec 18;9:1447. doi: 10.3389/fphar.2018.01447. eCollection 2018. Front Pharmacol. 2018. PMID: 30618744 Free PMC article.
-
Standardized Cirsium setidens Nakai Ethanolic Extract Suppresses Adipogenesis and Regulates Lipid Metabolisms in 3T3-L1 Adipocytes and C57BL/6J Mice Fed High-Fat Diets.J Med Food. 2017 Aug;20(8):763-776. doi: 10.1089/jmf.2017.3965. Epub 2017 Jul 7. J Med Food. 2017. PMID: 28686516
-
Bergamottin Inhibits Adipogenesis in 3T3-L1 Cells and Weight Regulation in Diet-Induced Obese Mice.Am J Chin Med. 2018;46(3):601-615. doi: 10.1142/S0192415X18500313. Epub 2018 Apr 4. Am J Chin Med. 2018. PMID: 29614883
-
5,7-Dimethoxyflavone Attenuates Obesity by Inhibiting Adipogenesis in 3T3-L1 Adipocytes and High-Fat Diet-Induced Obese C57BL/6J Mice.J Med Food. 2016 Dec;19(12):1111-1119. doi: 10.1089/jmf.2016.3800. Epub 2016 Nov 9. J Med Food. 2016. PMID: 27828718
-
Secoisolariciresinol diglucoside inhibits adipogenesis through the AMPK pathway.Eur J Pharmacol. 2018 Feb 5;820:235-244. doi: 10.1016/j.ejphar.2017.12.038. Epub 2017 Dec 18. Eur J Pharmacol. 2018. PMID: 29269018
Cited by
-
Sitagliptin reduces insulin resistance and improves rat liver steatosis via the SIRT1/AMPKα pathway.Exp Ther Med. 2018 Oct;16(4):3121-3128. doi: 10.3892/etm.2018.6554. Epub 2018 Aug 1. Exp Ther Med. 2018. PMID: 30214535 Free PMC article.
-
Pharmacokinetics and enterohepatic circulation of jervine, an antitumor steroidal alkaloid from Veratrum nigrum in rats.J Pharm Anal. 2019 Oct;9(5):367-372. doi: 10.1016/j.jpha.2019.04.004. Epub 2019 Apr 25. J Pharm Anal. 2019. PMID: 31929946 Free PMC article.
-
Phytochemical investigation on the aerial parts of Veratrum versicolor f. viride Nakai and their biological activities.Turk J Chem. 2023 Sep 12;47(6):1346-1354. doi: 10.55730/1300-0527.3618. eCollection 2023. Turk J Chem. 2023. PMID: 38544705 Free PMC article.
-
Review: Veratrum californicum Alkaloids.Molecules. 2021 Sep 30;26(19):5934. doi: 10.3390/molecules26195934. Molecules. 2021. PMID: 34641477 Free PMC article. Review.
-
UPLC-MS/MS Method for the Simultaneous Quantification of Eight Compounds in Rat Plasma and Its Application to a Pharmacokinetic Study after Oral Administration of Veratrum (Veratrum nigrum L.) Extract.Biomed Res Int. 2020 Oct 19;2020:8883277. doi: 10.1155/2020/8883277. eCollection 2020. Biomed Res Int. 2020. PMID: 33134392 Free PMC article.
References
-
- WHO. WHO Media Centre Fact Sheets: Obesity and Overweight. 2015, http://www.who.int/mediacentre/factsheets/fs311/en/
-
- Kopelman P. G. Obesity as a medical problem. Nature. 2000;404(6778):635–643. - PubMed
-
- Ntambi J. M., Kim Y.-C. Adipocyte differentiation and gene expression. The Journal of Nutrition. 2000;130(12):3122S–3126S. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

