Cancer Stem Cell-Secreted Macrophage Migration Inhibitory Factor Stimulates Myeloid Derived Suppressor Cell Function and Facilitates Glioblastoma Immune Evasion
- PMID: 27145382
- PMCID: PMC5820763
- DOI: 10.1002/stem.2393
Cancer Stem Cell-Secreted Macrophage Migration Inhibitory Factor Stimulates Myeloid Derived Suppressor Cell Function and Facilitates Glioblastoma Immune Evasion
Abstract
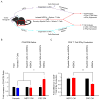
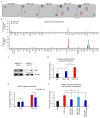
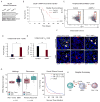
Shifting the balance away from tumor-mediated immune suppression toward tumor immune rejection is the conceptual foundation for a variety of immunotherapy efforts currently being tested. These efforts largely focus on activating antitumor immune responses but are confounded by multiple immune cell populations, including myeloid-derived suppressor cells (MDSCs), which serve to suppress immune system function. We have identified immune-suppressive MDSCs in the brains of GBM patients and found that they were in close proximity to self-renewing cancer stem cells (CSCs). MDSCs were selectively depleted using 5-flurouracil (5-FU) in a low-dose administration paradigm, which resulted in prolonged survival in a syngeneic mouse model of glioma. In coculture studies, patient-derived CSCs but not nonstem tumor cells selectively drove MDSC-mediated immune suppression. A cytokine screen revealed that CSCs secreted multiple factors that promoted this activity, including macrophage migration inhibitory factor (MIF), which was produced at high levels by CSCs. Addition of MIF increased production of the immune-suppressive enzyme arginase-1 in MDSCs in a CXCR2-dependent manner, whereas blocking MIF reduced arginase-1 production. Similarly to 5-FU, targeting tumor-derived MIF conferred a survival advantage to tumor-bearing animals and increased the cytotoxic T cell response within the tumor. Importantly, tumor cell proliferation, survival, and self-renewal were not impacted by MIF reduction, demonstrating that MIF is primarily an indirect promoter of GBM progression, working to suppress immune rejection by activating and protecting immune suppressive MDSCs within the GBM tumor microenvironment. Stem Cells 2016;34:2026-2039.
Keywords: Cancer stem cells; Glioblastoma; Immunotherapy; MIF; Macrophage migration inhibitory factor; Myeloid-derived suppressor cells; Tumor immune suppression; Tumor microenvironment.
© 2016 AlphaMed Press.
Conflict of interest statement
The authors declare no potential conflicts of interests.
Figures



Similar articles
-
Glioblastoma Myeloid-Derived Suppressor Cell Subsets Express Differential Macrophage Migration Inhibitory Factor Receptor Profiles That Can Be Targeted to Reduce Immune Suppression.Front Immunol. 2020 Jun 18;11:1191. doi: 10.3389/fimmu.2020.01191. eCollection 2020. Front Immunol. 2020. PMID: 32625208 Free PMC article.
-
Elevated levels of polymorphonuclear myeloid-derived suppressor cells in patients with glioblastoma highly express S100A8/9 and arginase and suppress T cell function.Neuro Oncol. 2016 Sep;18(9):1253-64. doi: 10.1093/neuonc/now034. Epub 2016 Mar 22. Neuro Oncol. 2016. PMID: 27006175 Free PMC article.
-
Modulating glioma-mediated myeloid-derived suppressor cell development with sulforaphane.PLoS One. 2017 Jun 30;12(6):e0179012. doi: 10.1371/journal.pone.0179012. eCollection 2017. PLoS One. 2017. PMID: 28666020 Free PMC article.
-
Explicating the Pivotal Pathogenic, Diagnostic, and Therapeutic Biomarker Potentials of Myeloid-Derived Suppressor Cells in Glioblastoma.Dis Markers. 2020 Nov 4;2020:8844313. doi: 10.1155/2020/8844313. eCollection 2020. Dis Markers. 2020. PMID: 33204365 Free PMC article. Review.
-
Immature myeloid cells in the tumor microenvironment: Implications for immunotherapy.Clin Immunol. 2018 Apr;189:34-42. doi: 10.1016/j.clim.2016.10.008. Epub 2016 Oct 21. Clin Immunol. 2018. PMID: 27777083 Free PMC article. Review.
Cited by
-
Specific N-cadherin-dependent pathways drive human breast cancer dormancy in bone marrow.Life Sci Alliance. 2021 Jun 2;4(7):e202000969. doi: 10.26508/lsa.202000969. Print 2021 Jul. Life Sci Alliance. 2021. PMID: 34078741 Free PMC article.
-
Pharmacological targeting of the tumor-immune symbiosis in glioblastoma.Trends Pharmacol Sci. 2022 Aug;43(8):686-700. doi: 10.1016/j.tips.2022.04.002. Epub 2022 May 7. Trends Pharmacol Sci. 2022. PMID: 35534356 Free PMC article. Review.
-
Recent Advances in Cancer Stem Cell-Targeted Immunotherapy.Cancers (Basel). 2019 Mar 5;11(3):310. doi: 10.3390/cancers11030310. Cancers (Basel). 2019. PMID: 30841635 Free PMC article. Review.
-
Glioblastoma stem cells: lessons from the tumor hierarchy in a lethal cancer.Genes Dev. 2019 Jun 1;33(11-12):591-609. doi: 10.1101/gad.324301.119. Genes Dev. 2019. PMID: 31160393 Free PMC article. Review.
-
Toca 511 gene transfer and treatment with the prodrug, 5-fluorocytosine, promotes durable antitumor immunity in a mouse glioma model.Neuro Oncol. 2017 Jul 1;19(7):930-939. doi: 10.1093/neuonc/nox037. Neuro Oncol. 2017. PMID: 28387849 Free PMC article.
References
-
- Stupp R, Mason WP, van den Bent MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352:987–996. - PubMed
-
- Furnari FB, Fenton T, Bachoo RM, et al. Malignant astrocytic glioma: Genetics, biology, and paths to treatment. Gene Dev. 2007;21:2683–2710. - PubMed
-
- Wen PY, Kesari S. Malignant gliomas in adults. N Engl J Med. 2008;359:492–507. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

