The Exocyst Complex in Health and Disease
- PMID: 27148529
- PMCID: PMC4828438
- DOI: 10.3389/fcell.2016.00024
The Exocyst Complex in Health and Disease
Abstract
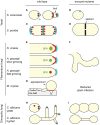
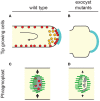
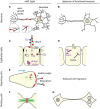
Exocytosis involves the fusion of intracellular secretory vesicles with the plasma membrane, thereby delivering integral membrane proteins to the cell surface and releasing material into the extracellular space. Importantly, exocytosis also provides a source of lipid moieties for membrane extension. The tethering of the secretory vesicle before docking and fusion with the plasma membrane is mediated by the exocyst complex, an evolutionary conserved octameric complex of proteins. Recent findings indicate that the exocyst complex also takes part in other intra-cellular processes besides secretion. These various functions seem to converge toward defining a direction of membrane growth in a range of systems from fungi to plants and from neurons to cilia. In this review we summarize the current knowledge of exocyst function in cell polarity, signaling and cell-cell communication and discuss implications for plant and animal health and disease.
Keywords: exocyst complex; fungi; mammals; pathogens; plants.
Figures
Similar articles
-
The exocyst complex in neurological disorders.Hum Genet. 2023 Aug;142(8):1263-1270. doi: 10.1007/s00439-023-02558-w. Epub 2023 Apr 22. Hum Genet. 2023. PMID: 37085629 Free PMC article. Review.
-
Exocyst and autophagy-related membrane trafficking in plants.J Exp Bot. 2017 Dec 18;69(1):47-57. doi: 10.1093/jxb/erx363. J Exp Bot. 2017. PMID: 29069430 Review.
-
The exocyst complex controls multiple events in the pathway of regulated exocytosis.Elife. 2024 Nov 25;12:RP92404. doi: 10.7554/eLife.92404. Elife. 2024. PMID: 39585321 Free PMC article.
-
The exocyst complex is an evolutionary battleground in plant-microbe interactions.Curr Opin Plant Biol. 2023 Dec;76:102482. doi: 10.1016/j.pbi.2023.102482. Epub 2023 Nov 3. Curr Opin Plant Biol. 2023. PMID: 37924562 Review.
-
The EXO70 inhibitor Endosidin2 alters plasma membrane protein composition in Arabidopsis roots.Front Plant Sci. 2023 May 31;14:1171957. doi: 10.3389/fpls.2023.1171957. eCollection 2023. Front Plant Sci. 2023. PMID: 37324680 Free PMC article.
Cited by
-
Necessity of rice resistance to planthoppers for OsEXO70H3 regulating SAMSL excretion and lignin deposition in cell walls.New Phytol. 2022 May;234(3):1031-1046. doi: 10.1111/nph.18012. Epub 2022 Feb 26. New Phytol. 2022. PMID: 35119102 Free PMC article.
-
Lung Endothelial Transcytosis.Compr Physiol. 2020 Mar 12;10(2):491-508. doi: 10.1002/cphy.c190012. Compr Physiol. 2020. PMID: 32163197 Free PMC article. Review.
-
The exocyst complex in neurological disorders.Hum Genet. 2023 Aug;142(8):1263-1270. doi: 10.1007/s00439-023-02558-w. Epub 2023 Apr 22. Hum Genet. 2023. PMID: 37085629 Free PMC article. Review.
-
Septins function in exocytosis via physical interactions with the exocyst complex in fission yeast cytokinesis.bioRxiv [Preprint]. 2025 Jul 1:2024.07.09.602728. doi: 10.1101/2024.07.09.602728. bioRxiv. 2025. PMID: 39026698 Free PMC article. Preprint.
-
The exocyst complex is required for developmental and regenerative neurite growth in vivo.Dev Biol. 2022 Dec;492:1-13. doi: 10.1016/j.ydbio.2022.09.005. Epub 2022 Sep 24. Dev Biol. 2022. PMID: 36162553 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

