Combined therapy with oncolytic adenoviruses encoding TRAIL and IL-12 genes markedly suppressed human hepatocellular carcinoma both in vitro and in an orthotopic transplanted mouse model
- PMID: 27154307
- PMCID: PMC4859966
- DOI: 10.1186/s13046-016-0353-8
Combined therapy with oncolytic adenoviruses encoding TRAIL and IL-12 genes markedly suppressed human hepatocellular carcinoma both in vitro and in an orthotopic transplanted mouse model
Erratum in
-
Erratum to: Combined therapy with oncolytic adenoviruses encoding TRAIL and IL-12 genes markedly suppressed human hepatocellular carcinoma both in vitro and in an orthotopic transplanted mouse model.J Exp Clin Cancer Res. 2016 Jun 17;35(1):96. doi: 10.1186/s13046-016-0365-4. J Exp Clin Cancer Res. 2016. PMID: 27316948 Free PMC article. No abstract available.
Abstract
Background: Gene-based virotherapy mediated by oncolytic viruses is currently experiencing a renaissance in cancer therapy. However, relatively little attention has been given to the potentiality of dual gene virotherapy strategy as a novel therapeutic approach to mediate triplex anticancer combination effects, particularly if the two suitable genes are well chosen. Both tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) and interleukin-12 (IL-12) have been emerged as promising pharmacological candidates in cancer therapy; however, the combined efficacy of TRAIL and IL-12 genes for treatment of human hepatocellular carcinoma (HCC) remains to be determined.
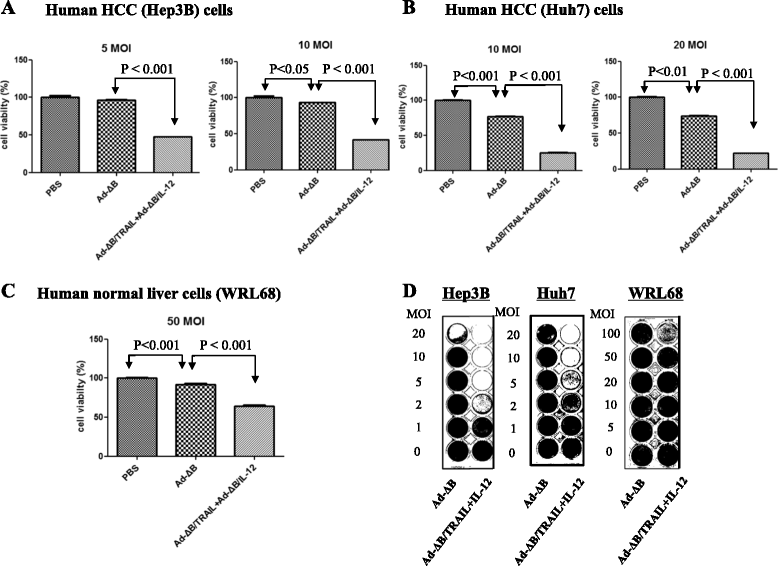
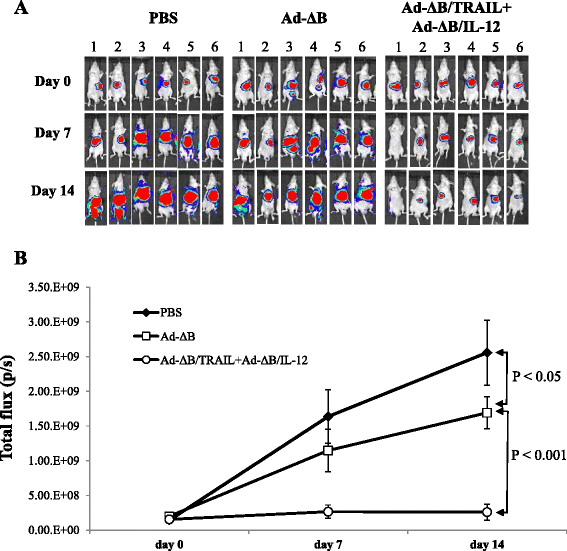
Methods: Herein, we investigated the therapeutic efficacy of concurrent therapy with two armed oncolytic adenoviruses encoding human TRAIL gene (Ad-ΔB/TRAIL) and IL-12 gene (Ad-ΔB/IL-12), respectively, on preclinical models of human HCC, and also elucidated the possible underlying mechanisms. The effects of Ad-ΔB/TRAIL+Ad-ΔB/IL-12 combination therapy were assessed both in vitro on Hep3B and HuH7 human HCC cell lines and in vivo on HCC-orthotopic model established in the livers of athymic nude mice by intrahepatic implantation of human Hep3B cells.
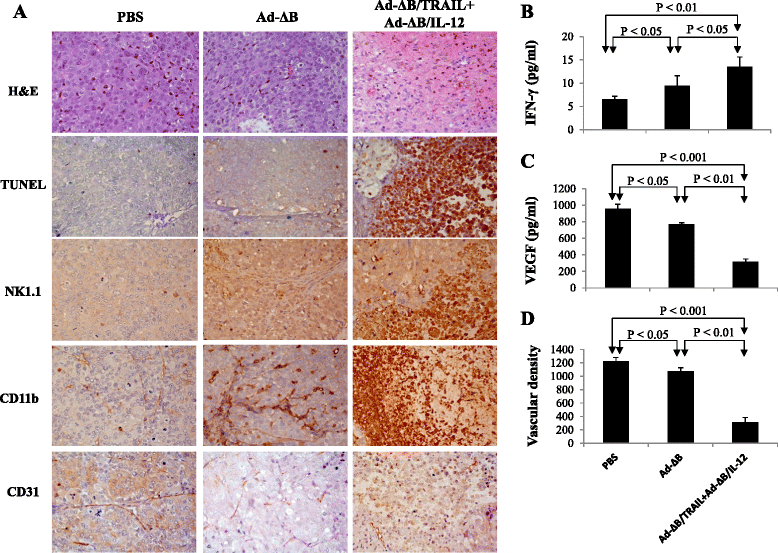
Results: Compared to therapy with non-armed control Ad-ΔB, combined therapy with Ad-ΔB/TRAIL+Ad-ΔB/IL-12 elicited profound anti-HCC killing effects on Hep3B and HuH7 cells and on the transplanted Hep3B-orthotopic model. Efficient viral replication and TRAIL and IL-12 expression were also confirmed in HCC cells and the harvested tumor tissues treated with this combination therapy. Mechanistically, co-therapy with Ad-ΔB/TRAIL+Ad-ΔB/IL-12 exhibited an enhanced effect on apoptosis promotion, activation of caspase-3 and-8, generation of anti-tumor immune response evidenced by upregulation of interferon gamma (IFN-γ) production and infiltration of natural killer-and antigen presenting cells, and remarkable repression of intratumor vascular endothelial growth factor (VEGF) and cluster of differentiation 31 (CD31) expression and tumor microvessel density.
Conclusions: Overall, our data showed a favorable therapeutic effect of Ad-ΔB/TRAIL+Ad-ΔB/IL-12 combination therapy against human HCC, and may therefore constitute a promising and effective therapeutic strategy for treating human HCC. However, further studies are warranted for its reliable clinical translation.
Keywords: Dual gene virotherapy; Hepatocellular carcinoma; Interleukin-12; Oncolytic adenoviruses; Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL).
Figures




Similar articles
-
Efficacy of combining ING4 and TRAIL genes in cancer-targeting gene virotherapy strategy: first evidence in preclinical hepatocellular carcinoma.Gene Ther. 2018 Jan;25(1):54-65. doi: 10.1038/gt.2017.86. Epub 2017 Sep 19. Gene Ther. 2018. PMID: 28925992 Free PMC article.
-
Gene-viro-therapy targeting liver cancer by a dual-regulated oncolytic adenoviral vector harboring IL-24 and TRAIL.Cancer Gene Ther. 2012 Jan;19(1):49-57. doi: 10.1038/cgt.2011.67. Epub 2011 Oct 7. Cancer Gene Ther. 2012. PMID: 21979578
-
Augmenting the antitumor effect of TRAIL by SOCS3 with double-regulated replicating oncolytic adenovirus in hepatocellular carcinoma.Hum Gene Ther. 2011 Sep;22(9):1109-19. doi: 10.1089/hum.2010.219. Epub 2011 Apr 21. Hum Gene Ther. 2011. PMID: 21361790
-
Recent advances in oncolytic adenovirus therapies for cancer.Curr Opin Virol. 2016 Dec;21:9-15. doi: 10.1016/j.coviro.2016.06.009. Epub 2016 Jul 2. Curr Opin Virol. 2016. PMID: 27379906 Free PMC article. Review.
-
Cellular genetic tools to control oncolytic adenoviruses for virotherapy of cancer.J Mol Med (Berl). 2008 Apr;86(4):363-77. doi: 10.1007/s00109-007-0291-1. Epub 2007 Dec 19. J Mol Med (Berl). 2008. PMID: 18214411 Review.
Cited by
-
IL-12 Family Cytokines in Cancer and Immunotherapy.Cancers (Basel). 2021 Jan 6;13(2):167. doi: 10.3390/cancers13020167. Cancers (Basel). 2021. PMID: 33418929 Free PMC article. Review.
-
Elevated MTSS1 expression associated with metastasis and poor prognosis of residual hepatitis B-related hepatocellular carcinoma.J Exp Clin Cancer Res. 2016 May 26;35(1):85. doi: 10.1186/s13046-016-0361-8. J Exp Clin Cancer Res. 2016. PMID: 27230279 Free PMC article.
-
Oncolytic Virus Encoding a Master Pro-Inflammatory Cytokine Interleukin 12 in Cancer Immunotherapy.Cells. 2020 Feb 10;9(2):400. doi: 10.3390/cells9020400. Cells. 2020. PMID: 32050597 Free PMC article. Review.
-
CD317 Promotes the survival of cancer cells through apoptosis-inducing factor.J Exp Clin Cancer Res. 2016 Jul 22;35(1):117. doi: 10.1186/s13046-016-0391-2. J Exp Clin Cancer Res. 2016. PMID: 27444183 Free PMC article.
-
Erratum to: Combined therapy with oncolytic adenoviruses encoding TRAIL and IL-12 genes markedly suppressed human hepatocellular carcinoma both in vitro and in an orthotopic transplanted mouse model.J Exp Clin Cancer Res. 2016 Jun 17;35(1):96. doi: 10.1186/s13046-016-0365-4. J Exp Clin Cancer Res. 2016. PMID: 27316948 Free PMC article. No abstract available.
References
-
- Stotz M, Gerger A, Haybaeck J, Kiesslich T, Bullock MD, Pichler M. Molecular targeted therapies in hepatocellular carcinoma: past, present and future. Anticancer Res. 2015;35(11):5737–44. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

