Degradation of oxidized proteins by the proteasome: Distinguishing between the 20S, 26S, and immunoproteasome proteolytic pathways
- PMID: 27155164
- PMCID: PMC4967006
- DOI: 10.1016/j.mam.2016.05.001
Degradation of oxidized proteins by the proteasome: Distinguishing between the 20S, 26S, and immunoproteasome proteolytic pathways
Abstract
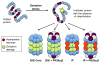
The proteasome is a ubiquitous and highly plastic multi-subunit protease with multi-catalytic activity that is conserved in all eukaryotes. The most widely known function of the proteasome is protein degradation through the 26S ubiquitin-proteasome system, responsible for the vast majority of protein degradation during homeostasis. However, the proteasome also plays an important role in adaptive immune responses and adaptation to oxidative stress. The unbound 20S proteasome, the core common to all proteasome conformations, is the main protease responsible for degrading oxidized proteins. During periods of acute stress, the 19S regulatory cap of the 26S proteasome disassociates from the proteolytic core, allowing for immediate ATP/ubiquitin-independent protein degradation by the 20S proteasome. Despite the abundance of unbound 20S proteasome compared to other proteasomal conformations, many publications fail to distinguish between the two proteolytic systems and often regard the 26S proteasome as the dominant protease. Further confounding the issue are the differential roles these two proteolytic systems have in adaptation and aging. In this review, we will summarize the increasing evidence that the 20S core proteasome constitutes the major conformation of the proteasome system and that it is far from a latent protease requiring activation by binding regulators.
Keywords: Adaptation; Immunoproteasome; Nrf2; Oxidative Stress; Proteasome; Proteolysis.
Copyright © 2016 Elsevier Ltd. All rights reserved.
Figures



Similar articles
-
Degradation of oxidized proteins by the 20S proteasome.Biochimie. 2001 Mar-Apr;83(3-4):301-10. doi: 10.1016/s0300-9084(01)01250-0. Biochimie. 2001. PMID: 11295490 Review.
-
Regulating the 20S proteasome ubiquitin-independent degradation pathway.Biomolecules. 2014 Sep 23;4(3):862-84. doi: 10.3390/biom4030862. Biomolecules. 2014. PMID: 25250704 Free PMC article. Review.
-
Ubiquitin-proteasome pathway and cellular responses to oxidative stress.Free Radic Biol Med. 2011 Jul 1;51(1):5-16. doi: 10.1016/j.freeradbiomed.2011.03.031. Epub 2011 Apr 8. Free Radic Biol Med. 2011. PMID: 21530648 Free PMC article. Review.
-
The proteasome and its role in the degradation of oxidized proteins.IUBMB Life. 2008 Nov;60(11):743-52. doi: 10.1002/iub.114. IUBMB Life. 2008. PMID: 18636510 Review.
-
Redox regulation of the proteasome via S-glutathionylation.Redox Biol. 2013 Dec 14;2:44-51. doi: 10.1016/j.redox.2013.12.003. Redox Biol. 2013. PMID: 24396728 Free PMC article. Review.
Cited by
-
Reduced Proteasome Activity and Enhanced Autophagy in Blood Cells of Psoriatic Patients.Int J Mol Sci. 2020 Oct 14;21(20):7608. doi: 10.3390/ijms21207608. Int J Mol Sci. 2020. PMID: 33066703 Free PMC article.
-
The insulin-degrading enzyme is an allosteric modulator of the 20S proteasome and a potential competitor of the 19S.Cell Mol Life Sci. 2018 Sep;75(18):3441-3456. doi: 10.1007/s00018-018-2807-y. Epub 2018 Mar 28. Cell Mol Life Sci. 2018. PMID: 29594388 Free PMC article.
-
Female Mice Reaching Exceptionally High Old Age Have Preserved 20S Proteasome Activities.Antioxidants (Basel). 2021 Aug 31;10(9):1397. doi: 10.3390/antiox10091397. Antioxidants (Basel). 2021. PMID: 34573029 Free PMC article.
-
Proteasome regulation of petite-negativity in fission yeast.bioRxiv [Preprint]. 2024 May 14:2024.05.09.593392. doi: 10.1101/2024.05.09.593392. bioRxiv. 2024. PMID: 38798546 Free PMC article. Preprint.
-
Genetic ablation of Lmp2 increases the susceptibility for impaired cardiac function.Front Mol Biosci. 2024 Mar 7;11:1148948. doi: 10.3389/fmolb.2024.1148948. eCollection 2024. Front Mol Biosci. 2024. PMID: 38516190 Free PMC article.
References
-
- Adams J, Kauffman M. Development of the proteasome inhibitor Velcade™ (Bortezomib) Cancer Invest. 2004;22:304–311. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

