Roles for retrotransposon insertions in human disease
- PMID: 27158268
- PMCID: PMC4859970
- DOI: 10.1186/s13100-016-0065-9
Roles for retrotransposon insertions in human disease
Abstract
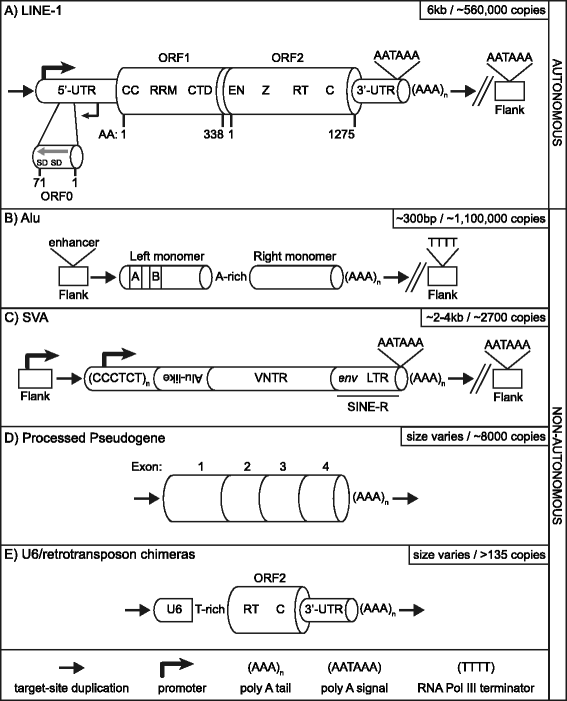
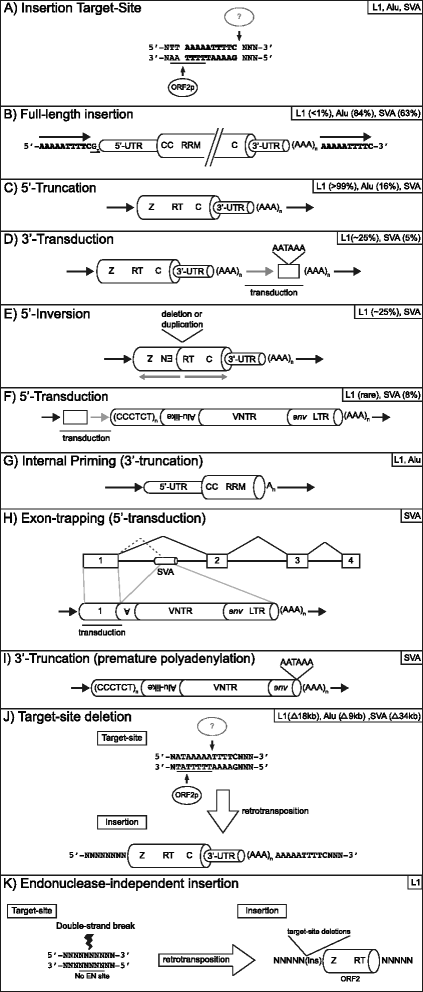
Over evolutionary time, the dynamic nature of a genome is driven, in part, by the activity of transposable elements (TE) such as retrotransposons. On a shorter time scale it has been established that new TE insertions can result in single-gene disease in an individual. In humans, the non-LTR retrotransposon Long INterspersed Element-1 (LINE-1 or L1) is the only active autonomous TE. In addition to mobilizing its own RNA to new genomic locations via a "copy-and-paste" mechanism, LINE-1 is able to retrotranspose other RNAs including Alu, SVA, and occasionally cellular RNAs. To date in humans, 124 LINE-1-mediated insertions which result in genetic diseases have been reported. Disease causing LINE-1 insertions have provided a wealth of insight and the foundation for valuable tools to study these genomic parasites. In this review, we provide an overview of LINE-1 biology followed by highlights from new reports of LINE-1-mediated genetic disease in humans.
Keywords: Alu; Autoimmunity; Cancer; Disease; LINE-1; Retrotransposition; Retrotransposon; SVA.
Figures
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

