Mesenchymal Stromal Cell-Derived Extracellular Vesicles Protect the Fetal Brain After Hypoxia-Ischemia
- PMID: 27160705
- PMCID: PMC4878333
- DOI: 10.5966/sctm.2015-0197
Mesenchymal Stromal Cell-Derived Extracellular Vesicles Protect the Fetal Brain After Hypoxia-Ischemia
Abstract
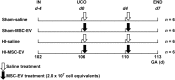
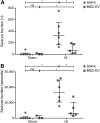
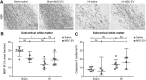
Preterm neonates are susceptible to perinatal hypoxic-ischemic brain injury, for which no treatment is available. In a preclinical animal model of hypoxic-ischemic brain injury in ovine fetuses, we have demonstrated the neuroprotective potential of systemically administered mesenchymal stromal cells (MSCs). The mechanism of MSC treatment is unclear but suggested to be paracrine, through secretion of extracellular vesicles (EVs). Therefore, we investigated in this study the protective effects of mesenchymal stromal cell-derived extracellular vesicles (MSC-EVs) in a preclinical model of preterm hypoxic-ischemic brain injury. Ovine fetuses were subjected to global hypoxia-ischemia by transient umbilical cord occlusion, followed by in utero intravenous administration of MSC-EVs. The therapeutic effects of MSC-EV administration were assessed by analysis of electrophysiological parameters and histology of the brain. Systemic administration of MSC-EVs improved brain function by reducing the total number and duration of seizures, and by preserving baroreceptor reflex sensitivity. These functional protections were accompanied by a tendency to prevent hypomyelination. Cerebral inflammation remained unaffected by the MSC-EV treatment. Our data demonstrate that MSC-EV treatment might provide a novel strategy to reduce the neurological sequelae following hypoxic-ischemic injury of the preterm brain. Our study results suggest that a cell-free preparation comprising neuroprotective MSC-EVs could substitute MSCs in the treatment of preterm neonates with hypoxic-ischemic brain injury, thereby circumventing the potential risks of systemic administration of living cells.
Significance: Bone marrow-derived mesenchymal stromal cells (MSCs) show promise in treating hypoxic-ischemic injury of the preterm brain. Study results suggest administration of extracellular vesicles, rather than intact MSCs, is sufficient to exert therapeutic effects and avoids potential concerns associated with administration of living cells. The therapeutic efficacy of systemically administered mesenchymal stromal cell-derived extracellular vesicles (MSC-EVs) on hypoxia-ischemia-induced injury was assessed in the preterm ovine brain. Impaired function and structural injury of the fetal brain was improved following global hypoxia-ischemia. A cell-free preparation of MSC-EVs could substitute for the cellular counterpart in the treatment of preterm neonates with hypoxic-ischemic brain injury. This may open new clinical applications for "off-the-shelf" interventions with MSC-EVs.
Keywords: Brain injury; Exosomes; Extracellular vesicles; Hypoxia-ischemia; Mesenchymal stromal cells; Preterm.
©AlphaMed Press.
Figures



References
-
- Fleiss B, Tann CJ, Degos V, et al. Inflammation-induced sensitization of the brain in term infants. Dev Med Child Neurol. 2015;57(suppl 3):17–28. - PubMed
-
- Azzopardi D, Robertson NJ, Cowan FM, et al. Pilot study of treatment with whole body hypothermia for neonatal encephalopathy. Pediatrics. 2000;106:684–694. - PubMed
-
- Briatore E, Ferrari F, Pomero G, et al. EEG findings in cooled asphyxiated newborns and correlation with site and severity of brain damage. Brain Dev. 2013;35:420–426. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

