Elucidation of the Biotransformation Pathways of a Galnac3-conjugated Antisense Oligonucleotide in Rats and Monkeys
- PMID: 27164023
- PMCID: PMC5014515
- DOI: 10.1038/mtna.2016.31
Elucidation of the Biotransformation Pathways of a Galnac3-conjugated Antisense Oligonucleotide in Rats and Monkeys
Abstract
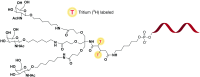
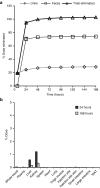
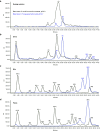
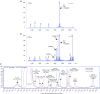
Triantennary N-acetyl galactosamine (GalNAc3) is a high-affinity ligand for hepatocyte-specific asialoglycoprotein receptors. Conjugation with GalNAc3 via a trishexylamino (THA)-C6 cluster significantly enhances antisense oligonucleotide (ASO) potency. Herein, the biotransformation, disposition, and elimination of the THA cluster of ION-681257, a GalNAc3-conjugated ASO currently in clinical development, are investigated in rats and monkey. Rats were administered a single subcutaneous dose of (3)H-radiolabeled ((3)H placed in THA) or nonradiolabeled ION-681257. Mass balance included radiometric profiling and metabolite fractionation with characterization by mass spectrometry. GalNAc3-conjugated ASOs were extensively distributed into liver. The THA-C6 triantenerrary GalNAc3 conjugate at the 5'-end of the ASO was rapidly metabolized and excreted with 25.67 ± 1.635% and 71.66 ± 4.17% of radioactivity recovered in urine and feces within 48 hours postdose. Unchanged drug, short-mer ASOs, and linker metabolites were detected in urine. Collectively, 14 novel linker associated metabolites were discovered including oxidation at each branching arm, initially by monooxidation at the β-position followed by dioxidation at the α-arm, and lastly, tri and tetra oxidations on the two remaining β-arms. Metabolites in bile and feces were identical to urine except for oxidized linear and cyclic linker metabolites. Enzymatic reaction phenotyping confirmed involvement of N-acetyl-β-glucosaminidase, deoxyribonuclease II, alkaline phosphatase, and alcohol + aldehyde dehydrogenases on the complex metabolism pathway for THA supplementing in vivo findings. Lastly, excreta from monkeys treated with ION-681257 revealed the identical series as observed in rat. In summary, our findings provide an improved understanding of GalNAc3-conjugated-ASO metabolism pathways which facilitate similar development programs.
Figures



Similar articles
-
Safety, Pharmacokinetic, and Pharmacodynamic Evaluation of a 2'-(2-Methoxyethyl)-D-ribose Antisense Oligonucleotide-Triantenarry N-Acetyl-galactosamine Conjugate that Targets the Human Transmembrane Protease Serine 6.J Pharmacol Exp Ther. 2021 Apr;377(1):51-63. doi: 10.1124/jpet.120.000222. Epub 2021 Jan 11. J Pharmacol Exp Ther. 2021. PMID: 33431610
-
Disposition and Pharmacokinetics of a GalNAc3-Conjugated Antisense Oligonucleotide Targeting Human Lipoprotein (a) in Monkeys.Nucleic Acid Ther. 2016 Dec;26(6):372-380. doi: 10.1089/nat.2016.0623. Epub 2016 Aug 8. Nucleic Acid Ther. 2016. PMID: 27500733
-
Integrated Assessment of the Clinical Performance of GalNAc3-Conjugated 2'-O-Methoxyethyl Chimeric Antisense Oligonucleotides: I. Human Volunteer Experience.Nucleic Acid Ther. 2019 Feb;29(1):16-32. doi: 10.1089/nat.2018.0753. Epub 2018 Dec 20. Nucleic Acid Ther. 2019. PMID: 30570431 Free PMC article. Clinical Trial.
-
Pharmacokinetics and Clinical Pharmacology Considerations of GalNAc3-Conjugated Antisense Oligonucleotides.Expert Opin Drug Metab Toxicol. 2019 Jun;15(6):475-485. doi: 10.1080/17425255.2019.1621838. Epub 2019 May 30. Expert Opin Drug Metab Toxicol. 2019. PMID: 31144994 Review.
-
Antisense oligonucleotides: absorption, distribution, metabolism, and excretion.Expert Opin Drug Metab Toxicol. 2021 Nov;17(11):1281-1292. doi: 10.1080/17425255.2021.1992382. Epub 2021 Oct 22. Expert Opin Drug Metab Toxicol. 2021. PMID: 34643122 Review.
Cited by
-
Evolution of complexity in non-viral oligonucleotide delivery systems: from gymnotic delivery through bioconjugates to biomimetic nanoparticles.RNA Biol. 2022 Jan;19(1):1256-1275. doi: 10.1080/15476286.2022.2147278. RNA Biol. 2022. PMID: 36411594 Free PMC article. Review.
-
GalNAc Conjugation Attenuates the Cytotoxicity of Antisense Oligonucleotide Drugs in Renal Tubular Cells.Mol Ther Nucleic Acids. 2019 Mar 1;14:67-79. doi: 10.1016/j.omtn.2018.11.005. Epub 2018 Nov 20. Mol Ther Nucleic Acids. 2019. PMID: 30583097 Free PMC article.
-
Docosanoic acid conjugation to siRNA enables functional and safe delivery to skeletal and cardiac muscles.Mol Ther. 2021 Apr 7;29(4):1382-1394. doi: 10.1016/j.ymthe.2020.12.023. Epub 2020 Dec 19. Mol Ther. 2021. PMID: 33348054 Free PMC article.
-
Efficient Liver Targeting by Polyvalent Display of a Compact Ligand for the Asialoglycoprotein Receptor.J Am Chem Soc. 2017 Mar 8;139(9):3528-3536. doi: 10.1021/jacs.6b12964. Epub 2017 Feb 23. J Am Chem Soc. 2017. PMID: 28230359 Free PMC article.
-
Biotransformation novel advances - 2021 year in review.Drug Metab Rev. 2022 Aug;54(3):207-245. doi: 10.1080/03602532.2022.2097253. Epub 2022 Aug 30. Drug Metab Rev. 2022. PMID: 35815654 Free PMC article. Review.
References
-
- Crooke, ST (1999). Molecular mechanisms of action of antisense drugs. Biochim Biophys Acta 1489: 31–44. - PubMed
-
- Bennett, CF and Swayze, EE (2010). RNA targeting therapeutics: molecular mechanisms of antisense oligonucleotides as a therapeutic platform. Annu Rev Pharmacol Toxicol 50: 259–293. - PubMed
-
- Kanasty, R, Dorkin, JR, Vegas, A and Anderson, D (2013). Delivery materials for siRNA therapeutics. Nat Mater 12: 967–977. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

