Minimal contribution of ERK1/2-MAPK signalling towards the maintenance of oncogenic GNAQQ209P-driven uveal melanomas in zebrafish
- PMID: 27166257
- PMCID: PMC5129960
- DOI: 10.18632/oncotarget.9207
Minimal contribution of ERK1/2-MAPK signalling towards the maintenance of oncogenic GNAQQ209P-driven uveal melanomas in zebrafish
Abstract
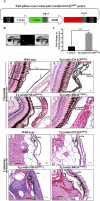
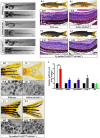
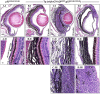
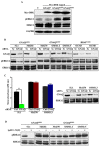
Mutations affecting Gαq proteins are pervasive in uveal melanoma (UM), suggesting they 'drive' UM pathogenesis. The ERK1/2-MAPK pathway is critical for cutaneous melanoma development and consequently an important therapeutic target. Defining the contribution of ERK1/2-MAPK signalling to UM development has been hampered by the lack of an informative animal model that spontaneously develops UM. Towards this end, we engineered transgenic zebrafish to express oncogenic GNAQQ209P in the melanocyte lineage. This resulted in hyperplasia of uveal melanocytes, but with no evidence of malignant progression, nor perturbation of skin melanocytes. Combining expression of oncogenic GNAQQ209P with p53 inactivation resulted in earlier onset and even more extensive hyperplasia of uveal melanocytes that progressed to UM. Immunohistochemistry revealed only weak immunoreactivity to phosphorylated (p)ERK1/2 in established uveal tumours-in contrast to strong immunoreactivity in oncogenic RAS-driven skin lesions-but ubiquitous positive staining for nuclear Yes-associated protein (YAP). Moreover, no changes were observed in pERK1/2 levels upon transient knockdown of GNAQ or phospholipase C-beta (PLC-β) inhibition in the majority of human UM cell lines we tested harbouring GNAQ mutations. In summary, our findings demonstrate a weak correlation between oncogenic GNAQQ209P mutation and sustained ERK1/2-MAPK activation, implying that ERK1/2 signalling is unlikely to be instrumental in the maintenance of GNAQQ209P-driven UMs.
Keywords: ERK1/2; GNAQ; p53; uveal melanoma; zebrafish.
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
RasGRP3 Mediates MAPK Pathway Activation in GNAQ Mutant Uveal Melanoma.Cancer Cell. 2017 May 8;31(5):685-696.e6. doi: 10.1016/j.ccell.2017.04.002. Cancer Cell. 2017. PMID: 28486107 Free PMC article.
-
Heterogeneity in Mitogen-Activated Protein Kinase (MAPK) Pathway Activation in Uveal Melanoma With Somatic GNAQ and GNA11 Mutations.Invest Ophthalmol Vis Sci. 2019 Jun 3;60(7):2474-2480. doi: 10.1167/iovs.18-26452. Invest Ophthalmol Vis Sci. 2019. PMID: 31173078 Free PMC article.
-
The protein kinase C inhibitor enzastaurin exhibits antitumor activity against uveal melanoma.PLoS One. 2012;7(1):e29622. doi: 10.1371/journal.pone.0029622. Epub 2012 Jan 12. PLoS One. 2012. PMID: 22253748 Free PMC article.
-
[Uveal Melanoma: Molecular and Genetic Mechanisms of Development and Therapeutic Approaches].Mol Biol (Mosk). 2024 Mar-Apr;58(2):189-203. Mol Biol (Mosk). 2024. PMID: 39355878 Review. Russian.
-
Exploring recent advances in signaling pathways and hallmarks of uveal melanoma: a comprehensive review.Explor Target Antitumor Ther. 2025 Apr 2;6:1002306. doi: 10.37349/etat.2025.1002306. eCollection 2025. Explor Target Antitumor Ther. 2025. PMID: 40177537 Free PMC article. Review.
Cited by
-
MITF deficiency accelerates GNAQ-driven uveal melanoma.Proc Natl Acad Sci U S A. 2022 May 10;119(19):e2107006119. doi: 10.1073/pnas.2107006119. Epub 2022 May 5. Proc Natl Acad Sci U S A. 2022. PMID: 35512098 Free PMC article.
-
GNAQ and GNA11 Genes: A Comprehensive Review on Oncogenesis, Prognosis and Therapeutic Opportunities in Uveal Melanoma.Cancers (Basel). 2022 Jun 22;14(13):3066. doi: 10.3390/cancers14133066. Cancers (Basel). 2022. PMID: 35804836 Free PMC article. Review.
-
The Melding of Drug Screening Platforms for Melanoma.Front Oncol. 2019 Jun 24;9:512. doi: 10.3389/fonc.2019.00512. eCollection 2019. Front Oncol. 2019. PMID: 31293965 Free PMC article. Review.
-
Genetic Engineering of Zebrafish in Cancer Research.Cancers (Basel). 2020 Aug 4;12(8):2168. doi: 10.3390/cancers12082168. Cancers (Basel). 2020. PMID: 32759814 Free PMC article. Review.
-
The Impact of Ultraviolet Radiation on the Aetiology and Development of Uveal Melanoma.Cancers (Basel). 2021 Apr 3;13(7):1700. doi: 10.3390/cancers13071700. Cancers (Basel). 2021. PMID: 33916693 Free PMC article. Review.
References
-
- Nathan P, Cohen V, Coupland S, Curtis K, Damato B, Evans J, Fenwick S, Kirkpatrick L, Li O, Marshall E, McGuirk K, Ottensmeier C, Pearce N, Salvi S, Stedman B, Szlosarek P, Turnbull N. Uveal Melanoma UK National Guidelines. Eur J Cancer. 2015;51:2404–2412. - PubMed
-
- Gray-Schopfer V, Wellbrock C, Marais R. Melanoma biology and new targeted therapy. Nature. 2007;445:851–857. - PubMed
-
- Zuidervaart W, van Nieuwpoort F, Stark M, Dijkman R, Packer L, Borgstein AM, Pavey S, van der Velden P, Out C, Jager MJ, Hayward NK, Gruis NA. Activation of the MAPK pathway is a common event in uveal melanomas although it rarely occurs through mutation of BRAF or RAS. Br J Cancer. 2005;92:2032–2038. - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

