Hypothalamic roles of mTOR complex I: integration of nutrient and hormone signals to regulate energy homeostasis
- PMID: 27166282
- PMCID: PMC4935144
- DOI: 10.1152/ajpendo.00121.2016
Hypothalamic roles of mTOR complex I: integration of nutrient and hormone signals to regulate energy homeostasis
Abstract
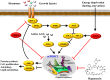
Mammalian or mechanistic target of rapamycin (mTOR) senses nutrient, energy, and hormone signals to regulate metabolism and energy homeostasis. mTOR activity in the hypothalamus, which is associated with changes in energy status, plays a critical role in the regulation of food intake and body weight. mTOR integrates signals from a variety of "energy balancing" hormones such as leptin, insulin, and ghrelin, although its action varies in response to these distinct hormonal stimuli as well as across different neuronal populations. In this review, we summarize and highlight recent findings regarding the functional roles of mTOR complex 1 (mTORC1) in the hypothalamus specifically in its regulation of body weight, energy expenditure, and glucose/lipid homeostasis. Understanding the role and underlying mechanisms behind mTOR-related signaling in the brain will undoubtedly pave new avenues for future therapeutics and interventions that can combat obesity, insulin resistance, and diabetes.
Keywords: energy homeostasis; hormones; hypothalamus; mTOR; nutrients.
Copyright © 2016 the American Physiological Society.
Figures

Similar articles
-
[Role of the mTOR pathway in the central regulation of energy balance].Biol Aujourdhui. 2015;209(4):295-307. doi: 10.1051/jbio/2016009. Epub 2016 Mar 28. Biol Aujourdhui. 2015. PMID: 27021048 Review. French.
-
Hypothalamic mTOR: the rookie energy sensor.Curr Mol Med. 2014 Jan;14(1):3-21. doi: 10.2174/1566524013666131118103706. Curr Mol Med. 2014. PMID: 24236459 Review.
-
Influence of mTOR in energy and metabolic homeostasis.Mol Cell Endocrinol. 2014 Nov;397(1-2):67-77. doi: 10.1016/j.mce.2014.07.015. Epub 2014 Aug 7. Mol Cell Endocrinol. 2014. PMID: 25109278 Review.
-
The role of hypothalamic mTORC1 signaling in insulin regulation of food intake, body weight, and sympathetic nerve activity in male mice.Endocrinology. 2015 Apr;156(4):1398-407. doi: 10.1210/en.2014-1660. Epub 2015 Jan 9. Endocrinology. 2015. PMID: 25574706 Free PMC article.
-
Hypothalamic carnitine metabolism integrates nutrient and hormonal feedback to regulate energy homeostasis.Mol Cell Endocrinol. 2015 Dec 15;418 Pt 1:9-16. doi: 10.1016/j.mce.2015.08.002. Epub 2015 Aug 8. Mol Cell Endocrinol. 2015. PMID: 26261054 Review.
Cited by
-
Metabolic Regulation of Hypoxia-Inducible Factors in Hypothalamus.Front Endocrinol (Lausanne). 2021 Mar 8;12:650284. doi: 10.3389/fendo.2021.650284. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 33763034 Free PMC article. Review.
-
Intestinal Flora Affect Alzheimer's Disease by Regulating Endogenous Hormones.Neurochem Res. 2022 Dec;47(12):3565-3582. doi: 10.1007/s11064-022-03784-w. Epub 2022 Oct 30. Neurochem Res. 2022. PMID: 36309938 Review.
-
Involvement of Mechanistic Target of Rapamycin (mTOR) in Valine Orexigenic Effects in Rainbow Trout.Aquac Nutr. 2022 Sep 27;2022:7509382. doi: 10.1155/2022/7509382. eCollection 2022. Aquac Nutr. 2022. PMID: 36860456 Free PMC article.
-
Genetic deletion of mast cell serotonin synthesis prevents the development of obesity and insulin resistance.Nat Commun. 2020 Jan 23;11(1):463. doi: 10.1038/s41467-019-14080-7. Nat Commun. 2020. PMID: 31974364 Free PMC article.
-
Metabolism Regulation and Redox State: Insight into the Role of Superoxide Dismutase 1.Int J Mol Sci. 2020 Sep 10;21(18):6606. doi: 10.3390/ijms21186606. Int J Mol Sci. 2020. PMID: 32927603 Free PMC article. Review.
References
-
- Aguilar V, Alliouachene S, Sotiropoulos A, Sobering A, Athea Y, Djouadi F, Miraux S, Thiaudiere E, Foretz M, Viollet B, Diolez P, Bastin J, Benit P, Rustin P, Carling D, Sandri M, Ventura-Clapier R, Pende M. S6 kinase deletion suppresses muscle growth adaptations to nutrient availability by activating AMP kinase. Cell Metab 5: 476–487, 2007. - PubMed
-
- Andre C, Cota D. Coupling nutrient sensing to metabolic homoeostasis: the role of the mammalian target of rapamycin complex 1 pathway. Proc Nutr Soc 71: 502–510, 2012. - PubMed
-
- Appenzeller-Herzog C, Hall MN. Bidirectional crosstalk between endoplasmic reticulum stress and mTOR signaling. Trends Cell Biol 22: 274–282, 2012. - PubMed
-
- Astrinidis A, Henske EP. Tuberous sclerosis complex: linking growth and energy signaling pathways with human disease. Oncogene 24: 7475–7481, 2005. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

