Application of iridium(III) complex in label-free and non-enzymatic electrochemical detection of hydrogen peroxide based on a novel "on-off-on" switch platform
- PMID: 27170211
- PMCID: PMC4864421
- DOI: 10.1038/srep25774
Application of iridium(III) complex in label-free and non-enzymatic electrochemical detection of hydrogen peroxide based on a novel "on-off-on" switch platform
Abstract
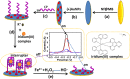
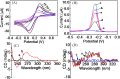
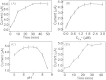
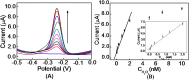
We herein report a label-free and non-enzymatic electrochemical sensor for the highly sensitive detection of hydrogen peroxide (H2O2) based on a novel "on-off-on" switch system. In our design, MB was used as an electron mediator to accelerate the electron transfer while AuNPs was used to amplify the electrochemical signal due to its excellent biocompatibility and good conductivity. The "switch-off" state was achieved by introducing the guanine-rich capture probe (CP) and an iridium complex onto the electrode surface to form a hydrophobic layer, which then hinders electron transfer. Upon addition of H2O2, fenton reaction occurs and produces OH• in the presence of Fe(2+). The OH• cleaves the CP into DNA fragments, thus resulting in the release of CP and iridium complex from the sensing interface, recovering the electrochemical signal to generate a "switch-on" state. Based on this novel switch system, a detection limit as low as 3.2 pM can be achieved for H2O2 detection. Moreover, satisfactory results were obtained by using this method for the detection of H2O2 in sterilized milk. To the best of our knowledge, this is the first G-quadruplex-based electrochemical sensor using an iridium(III) complex.
Figures
Similar articles
-
A cyclometalated iridium(III) complex used as a conductor for the electrochemical sensing of IFN-γ.Sci Rep. 2017 Feb 15;7:42740. doi: 10.1038/srep42740. Sci Rep. 2017. PMID: 28198433 Free PMC article.
-
G-quadruplex-selective iridium(III) complex as a novel electrochemiluminescence probe for switch-on assay of double-stranded DNA.Anal Bioanal Chem. 2022 May;414(13):3755-3763. doi: 10.1007/s00216-022-04018-2. Epub 2022 Apr 9. Anal Bioanal Chem. 2022. PMID: 35396609
-
Ultrasensitive electrochemical detection of miRNA-21 by using an iridium(III) complex as catalyst.Biosens Bioelectron. 2016 Dec 15;86:454-458. doi: 10.1016/j.bios.2016.07.001. Epub 2016 Jul 2. Biosens Bioelectron. 2016. PMID: 27424263
-
Au nanoparticles decorated C60 nanoparticle-based label-free electrochemiluminesence aptasensor via a novel "on-off-on" switch system.Biomaterials. 2015 Jun;52:476-83. doi: 10.1016/j.biomaterials.2015.02.058. Epub 2015 Mar 18. Biomaterials. 2015. PMID: 25818453
-
Zettomole electrochemical HIV DNA detection using 2D DNA-Au nanowire structure, hemin/G-quadruplex and polymerase chain reaction multi-signal synergistic amplification.Anal Chim Acta. 2021 May 15;1159:338428. doi: 10.1016/j.aca.2021.338428. Epub 2021 Mar 20. Anal Chim Acta. 2021. PMID: 33867042
Cited by
-
One-step Synthesized Silver Nanoparticles Using Isoimperatorin: Evaluation of Photocatalytic, and Electrochemical Activities.Sci Rep. 2020 Feb 4;10(1):1762. doi: 10.1038/s41598-020-58697-x. Sci Rep. 2020. PMID: 32020015 Free PMC article.
-
Nonenzymatic sensing of hydrogen peroxide using a glassy carbon electrode modified with graphene oxide, a polyamidoamine dendrimer, and with polyaniline deposited by the Fenton reaction.Mikrochim Acta. 2018 Dec 1;185(12):569. doi: 10.1007/s00604-018-3089-7. Mikrochim Acta. 2018. PMID: 30506518
-
A cyclometalated iridium(III) complex used as a conductor for the electrochemical sensing of IFN-γ.Sci Rep. 2017 Feb 15;7:42740. doi: 10.1038/srep42740. Sci Rep. 2017. PMID: 28198433 Free PMC article.
-
G-quadruplex-selective iridium(III) complex as a novel electrochemiluminescence probe for switch-on assay of double-stranded DNA.Anal Bioanal Chem. 2022 May;414(13):3755-3763. doi: 10.1007/s00216-022-04018-2. Epub 2022 Apr 9. Anal Bioanal Chem. 2022. PMID: 35396609
-
Editorial: Immunomodulatory role of metalloproteases in chronic inflammatory diseases.Front Immunol. 2023 Apr 11;14:1196791. doi: 10.3389/fimmu.2023.1196791. eCollection 2023. Front Immunol. 2023. PMID: 37114053 Free PMC article. No abstract available.
References
-
- Baghayeri M., Zare E. N. & Lakouraj M. M. A simple hydrogen peroxide biosensor based on a novel electro-magnetic poly(p-phenylenediamine)@Fe3O4 nanocomposite. Biosens. Bioelectron. 55, 259–265 (2014). - PubMed
-
- Chen X. et al. Advances in enzyme-free electrochemical sensors for hydrogen peroxide, glucose, and uric acid. Microchim. Acta 181, 689–705 (2014).
-
- Terai T. & Nagano T. Small-molecule fluorophores and fluorescent probes for bioimaging. Arch. Eur. Phys. 465, 347–359 (2013). - PubMed
-
- Liu Q. et al. NiO nanoparticles modified with 5,10,15,20-tetrakis(4-carboxyl pheyl)-porphyrin: Promising peroxidase mimetics for H2O2 and glucose detection. Biosens. Bioelectron. 64, 147–153 (2014). - PubMed
-
- Zhang Y. et al. Fabrication of 2D ordered mesoporous carbon nitride and its use as electrochemical sensing platform for H2O2, nitrobenzene, and NADH detection. Biosens. Bioelectron. 53, 250–256 (2014). - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

