Radical SAM catalysis via an organometallic intermediate with an Fe-[5'-C]-deoxyadenosyl bond
- PMID: 27174986
- PMCID: PMC4929858
- DOI: 10.1126/science.aaf5327
Radical SAM catalysis via an organometallic intermediate with an Fe-[5'-C]-deoxyadenosyl bond
Abstract
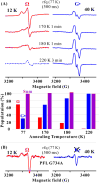
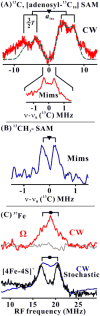
Radical S-adenosylmethionine (SAM) enzymes use a [4Fe-4S] cluster to cleave SAM to initiate diverse radical reactions. These reactions are thought to involve the 5'-deoxyadenosyl radical intermediate, which has not yet been detected. We used rapid freeze-quenching to trap a catalytically competent intermediate in the reaction catalyzed by the radical SAM enzyme pyruvate formate-lyase activating enzyme. Characterization of the intermediate by electron paramagnetic resonance and (13)C, (57)Fe electron nuclear double-resonance spectroscopies reveals that it contains an organometallic center in which the 5' carbon of a SAM-derived deoxyadenosyl moiety forms a bond with the unique iron site of the [4Fe-4S] cluster. Discovery of this intermediate extends the list of enzymatic bioorganometallic centers to the radical SAM enzymes, the largest enzyme superfamily known, and reveals intriguing parallels to B12 radical enzymes.
Copyright © 2016, American Association for the Advancement of Science.
Figures


Similar articles
-
Mechanism of Radical Initiation in the Radical S-Adenosyl-l-methionine Superfamily.Acc Chem Res. 2018 Nov 20;51(11):2611-2619. doi: 10.1021/acs.accounts.8b00356. Epub 2018 Oct 15. Acc Chem Res. 2018. PMID: 30346729 Free PMC article. Review.
-
Electron-nuclear double resonance spectroscopic evidence that S-adenosylmethionine binds in contact with the catalytically active [4Fe-4S](+) cluster of pyruvate formate-lyase activating enzyme.J Am Chem Soc. 2002 Mar 27;124(12):3143-51. doi: 10.1021/ja012034s. J Am Chem Soc. 2002. PMID: 11902903
-
Spectroscopic approaches to elucidating novel iron-sulfur chemistry in the "radical-Sam" protein superfamily.Inorg Chem. 2005 Feb 21;44(4):727-41. doi: 10.1021/ic0484811. Inorg Chem. 2005. PMID: 15859242
-
Paradigm Shift for Radical S-Adenosyl-l-methionine Reactions: The Organometallic Intermediate Ω Is Central to Catalysis.J Am Chem Soc. 2018 Jul 18;140(28):8634-8638. doi: 10.1021/jacs.8b04061. Epub 2018 Jul 6. J Am Chem Soc. 2018. PMID: 29954180 Free PMC article.
-
Radical SAM enzymes: Nature's choice for radical reactions.FEBS Lett. 2023 Jan;597(1):92-101. doi: 10.1002/1873-3468.14519. Epub 2022 Oct 27. FEBS Lett. 2023. PMID: 36251330 Free PMC article. Review.
Cited by
-
Organometallic Complex Formed by an Unconventional Radical S-Adenosylmethionine Enzyme.J Am Chem Soc. 2016 Aug 10;138(31):9755-8. doi: 10.1021/jacs.6b04155. Epub 2016 Aug 2. J Am Chem Soc. 2016. PMID: 27465315 Free PMC article.
-
Overall Retention of Methyl Stereochemistry during B12-Dependent Radical SAM Methyl Transfer in Fosfomycin Biosynthesis.Biochemistry. 2021 May 25;60(20):1587-1596. doi: 10.1021/acs.biochem.1c00113. Epub 2021 May 4. Biochemistry. 2021. PMID: 33942609 Free PMC article.
-
Radical S-adenosylmethionine maquette chemistry: Cx3Cx2C peptide coordinated redox active [4Fe-4S] clusters.J Biol Inorg Chem. 2019 Sep;24(6):793-807. doi: 10.1007/s00775-019-01708-8. Epub 2019 Sep 5. J Biol Inorg Chem. 2019. PMID: 31486952
-
Radical SAM enzymes: surprises along the path to understanding mechanism.J Biol Inorg Chem. 2019 Sep;24(6):769-776. doi: 10.1007/s00775-019-01706-w. Epub 2019 Sep 7. J Biol Inorg Chem. 2019. PMID: 31494759 Free PMC article. Review.
-
The B12-independent glycerol dehydratase activating enzyme from Clostridium butyricum cleaves SAM to produce 5'-deoxyadenosine and not 5'-deoxy-5'-(methylthio)adenosine.J Inorg Biochem. 2022 Feb;227:111662. doi: 10.1016/j.jinorgbio.2021.111662. Epub 2021 Nov 12. J Inorg Biochem. 2022. PMID: 34847521 Free PMC article.
References
-
- Sicoli G, et al. Fine-tuning of a radical-based reaction by radical S-adenosyl-L-methionine tryptophan lyase. Science. 2016;351:1320. - PubMed
-
- Walsby CJ, Ortillo D, Broderick WE, Broderick JB, Hoffman BM. An anchoring role for FeS Clusters: Chelation of the amino acid moiety of S-adenosylmethionine to the unique iron site of the [4Fe-4S] cluster of pyruvate formate-lyase activating enzyme. J Am Chem Soc. 2002;124:11270. - PubMed
-
- Chen D, Walsby C, Hoffman BM, Frey PA. Coordination and mechanism of reversible cleavage of S-adenosylmethionine by the [4Fe-4S] center in lysine 2,3-aminomutase. J Am Chem Soc. 2003;125:11788. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

