Parishin C's prevention of Aβ 1-42-induced inhibition of long-term potentiation is related to NMDA receptors
- PMID: 27175329
- PMCID: PMC4857013
- DOI: 10.1016/j.apsb.2016.03.009
Parishin C's prevention of Aβ 1-42-induced inhibition of long-term potentiation is related to NMDA receptors
Abstract
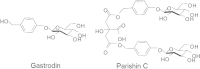
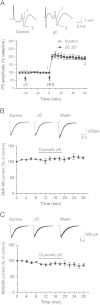
The rhizome of Gastrodia elata (GE), a herb medicine, has been used for treatment of neuronal disorders in Eastern Asia for hundreds of years. Parishin C is a major ingredient of GE. In this study, the i.c.v. injection of soluble Aβ 1-42 oligomers model of LTP injury was used. We investigated the effects of parishin C on the improvement of LTP in soluble Aβ 1-42 oligomer-injected rats and the underlying electrophysiological mechanisms. Parishin C (i.p. or i.c.v.) significantly ameliorated LTP impairment induced by i.c.v. injection of soluble Aβ 1-42 oligomers. In cultured hippocampal neurons, soluble Aβ 1-42 oligomers significantly inhibited NMDAR currents while not affecting AMPAR currents and voltage-dependent currents. Pretreatment with parishin C protected NMDA receptor currents from the damage induced by Aβ. In summary, parishin C improved LTP deficits induced by soluble Aβ 1-42 oligomers. The protection by parishin C against Aβ-induced LTP damage might be related to NMDA receptors.
Keywords: Ion channels; Long-term potentiation; NMDA receptors; Neuroprotection; Parishin C.
Figures



Similar articles
-
[Structure-activity relationship of gastrodin and parishins on learning and memory deficits induced by scopolamine].Yao Xue Xue Bao. 2016 May;51(5):743-8. Yao Xue Xue Bao. 2016. PMID: 29874020 Chinese.
-
Therapeutic significance of NR2B-containing NMDA receptors and mGluR5 metabotropic glutamate receptors in mediating the synaptotoxic effects of β-amyloid oligomers on long-term potentiation (LTP) in murine hippocampal slices.Neuropharmacology. 2011 May;60(6):982-90. doi: 10.1016/j.neuropharm.2011.01.051. Epub 2011 Feb 12. Neuropharmacology. 2011. PMID: 21310164
-
MRZ-99030 - A novel modulator of Aβ aggregation: II - Reversal of Aβ oligomer-induced deficits in long-term potentiation (LTP) and cognitive performance in rats and mice.Neuropharmacology. 2015 May;92:170-82. doi: 10.1016/j.neuropharm.2014.12.037. Epub 2015 Jan 28. Neuropharmacology. 2015. PMID: 25637092
-
The rhizome of Gastrodia elata Blume - An ethnopharmacological review.J Ethnopharmacol. 2016 Aug 2;189:361-85. doi: 10.1016/j.jep.2016.06.057. Epub 2016 Jul 1. J Ethnopharmacol. 2016. PMID: 27377337 Review.
-
[Effects of amyloid β-protein on hippocampal long-term potentiation].Sheng Li Xue Bao. 2010 Dec 25;62(6):479-88. Sheng Li Xue Bao. 2010. PMID: 21170492 Review. Chinese.
Cited by
-
Specific gut microbiome and metabolome changes in patients with continuous ambulatory peritoneal dialysis and comparison between patients with different dialysis vintages.Front Med (Lausanne). 2024 Jan 5;10:1302352. doi: 10.3389/fmed.2023.1302352. eCollection 2023. Front Med (Lausanne). 2024. PMID: 38249961 Free PMC article.
-
Sulfur-enriched alkaloids from the root of Isatis indigotica.Acta Pharm Sin B. 2018 Oct;8(6):933-943. doi: 10.1016/j.apsb.2018.08.005. Epub 2018 Aug 23. Acta Pharm Sin B. 2018. PMID: 30505662 Free PMC article.
-
The pathological roles of NDRG2 in Alzheimer's disease, a study using animal models and APPwt-overexpressed cells.CNS Neurosci Ther. 2017 Aug;23(8):667-679. doi: 10.1111/cns.12716. Epub 2017 Jul 2. CNS Neurosci Ther. 2017. PMID: 28670853 Free PMC article.
-
C19-Diterpenoid alkaloid arabinosides from an aqueous extract of the lateral root of Aconitum carmichaelii and their analgesic activities.Acta Pharm Sin B. 2018 May;8(3):409-419. doi: 10.1016/j.apsb.2018.03.009. Epub 2018 Mar 27. Acta Pharm Sin B. 2018. PMID: 29881680 Free PMC article.
-
Sleep promoting and omics exploration on probiotics fermented Gastrodia elata Blume.NPJ Sci Food. 2024 Jun 18;8(1):33. doi: 10.1038/s41538-024-00277-8. NPJ Sci Food. 2024. PMID: 38890318 Free PMC article.
References
-
- Zhu A.H., Tian J.Z., Zhong J., Yang C.Z., Shi J., Yin J.X. A clinical study on a randomized, double-blind control of Chinese medicine granules in treatment of vascular dementi. China J Chin Mater Med. 2006;31:1722–1725. - PubMed
-
- Xu WJ, Tan XC, Zhang M, Zhao LM, Liu Y, Shi JG, et al., inventor; Beijing Kelaibo Pharmaceutical Development, assignee. Gastrodia elata plant extract for preventing senile dementia and its preparing method. China Patent CN200510128785. 2007 June 13
-
- Wang K, Shi JG, Zhao LM, Zhao DL, Zou DC, Zhang M, et al., inventor; Beijing Collab Pharma Co. Ltd., assignee. Application of Gastrodiaelata blume parishin extractive in preparation of medicament for protecting brain. China Patent CN102727506A. 2012 October 17
-
- Wang K, Shi JG, Liu Y, Zhao LM, Zhang M, Chen MX, inventor; Beijing Collab Pharma Co. Ltd., assignee. Gastrodiaelata blume plant extract for preventing and treating vascular dementia and preparation method thereof. China Patent CN101628086A 2010 January 20
LinkOut - more resources
Full Text Sources
Other Literature Sources

