Bupropion therapy during pregnancy: the drug and its major metabolites in umbilical cord plasma and amniotic fluid
- PMID: 27180885
- PMCID: PMC5321070
- DOI: 10.1016/j.ajog.2016.05.016
Bupropion therapy during pregnancy: the drug and its major metabolites in umbilical cord plasma and amniotic fluid
Abstract
Background: Bupropion is used for treatment of depression during pregnancy. However, its use as a smoking cessation aid for pregnant women is currently under evaluation.
Objective: The aim of this opportunistic study was to investigate the transfer of bupropion and its major pharmacologically active metabolites, hydroxybupropion and threohydrobupropion, across the placenta in vivo. In addition, the concentrations of the drug and its metabolites were determined in the amniotic fluid.
Study design: The following samples were collected at deliveries from 22 women taking bupropion: maternal blood (n = 22), umbilical cord venous blood (n = 22), and amniotic fluid (n = 9). The concentrations of the drug and its metabolites in blood plasma and amniotic fluid were determined by means of liquid chromatography-mass spectrometry. Placental passage was calculated as a ratio of umbilical cord venous plasma to maternal plasma concentrations.
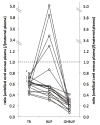
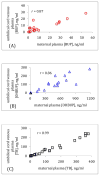
Results: The levels of hydroxybupropion and threohydrobupropion in umbilical cord venous plasma were invariably lower than their corresponding concentrations in maternal plasma. The concentrations of bupropion in umbilical cord plasma were lower than in maternal plasma in the majority of the maternal-cord blood pairs. The median values of the umbilical cord venous plasma to maternal plasma ratios were: bupropion, 0.53 (interquartile range 0.35, n = 18), hydroxybupropion, 0.21 (interquartile range 0.12, n = 18), and threohydrobupropion, 0.61 (interquartile range 0.11, n = 21). In umbilical cord venous plasma, the median concentration of bupropion was 5.3 ng/mL; hydroxybupropion, 103.6 ng/mL; and threohydrobupropion, 59.6 ng/mL. Bupropion and its metabolites were detectable in the amniotic fluid but the concentrations of threohydrobupropion were higher than those in the corresponding umbilical cord venous plasma.
Conclusion: Bupropion and its active metabolites cross the placenta to the fetal circulation. The concentrations of hydroxybupropion and threohydrobupropion in umbilical cord venous plasma were higher than bupropion concentrations suggesting a higher fetal exposure to the metabolites than the parent drug. The higher levels of threohydrobupropion in the amniotic fluid than those in umbilical cord venous plasma suggest that enzymes involved in the metabolism of bupropion to threohydrobupropion are most likely active in the fetus. The biological consequences of fetal exposure to maternally administered bupropion and/or its active metabolites via placental transfer and recirculation of the amniotic fluid are yet to be determined.
Keywords: amniotic fluid; bupropion; hydroxybupropion; placental passage; pregnancy; threohydrobupropion; umbilical cord plasma.
Copyright © 2016. Published by Elsevier Inc.
Conflict of interest statement
Figures
References
-
- Way CM. Safety of newer antidepressants in pregnancy. Pharmacotherapy. 2007 Apr;27(4):546–52. Review. - PubMed
-
- Patkar AA, Bilal L, Masand PS. Pharmacotherapy of depression in pregnancy. Ann Clin Psychiatry. 2004 Apr-Jun;16(2):87–100. Review. - PubMed
-
- Gentile S. The safety of newer antidepressants in pregnancy and breastfeeding. Drug Saf. 2005;28(2):137–52. Review. - PubMed
-
- Product monograph of Wellbutrin SR tablets. GlaxoSmithKline; North York: Jul, 2014.
-
- Product monograph of Wellbutrin XL tablets. Valeant Pharmaceutical International; Bridgewater, NJ: Oct, 2014. Reference ID: 2979024.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

