Dual Function of Ccr5 during Langat Virus Encephalitis: Reduction in Neutrophil-Mediated Central Nervous System Inflammation and Increase in T Cell-Mediated Viral Clearance
- PMID: 27183602
- PMCID: PMC4973474
- DOI: 10.4049/jimmunol.1502452
Dual Function of Ccr5 during Langat Virus Encephalitis: Reduction in Neutrophil-Mediated Central Nervous System Inflammation and Increase in T Cell-Mediated Viral Clearance
Abstract
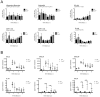
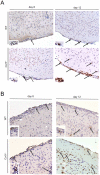
Tick-borne encephalitis virus (TBEV) is a vector-transmitted flavivirus that causes potentially fatal neurologic infection. There are thousands of cases reported annually, and despite the availability of an effective vaccine, the incidence of TBEV is increasing worldwide. Importantly, up to 30% of affected individuals develop long-term neurologic sequelae. We investigated the role of chemokine receptor Ccr5 in a mouse model of TBEV infection using the naturally attenuated tick-borne flavivirus Langat virus (LGTV). Ccr5-deficient mice presented with an increase in viral replication within the CNS and decreased survival during LGTV encephalitis compared with wild-type controls. This enhanced susceptibility was due to the temporal lag in lymphocyte migration into the CNS. Adoptive transfer of wild-type T cells, but not Ccr5-deficient T cells, significantly improved survival outcome in LGTV-infected Ccr5-deficient mice. Concomitantly, a significant increase in neutrophil migration into the CNS in LGTV-infected Ccr5(-/-) mice was documented at the late stage of infection. Ab-mediated depletion of neutrophils in Ccr5(-/-) mice resulted in a significant improvement in mortality, a decrease in viral load, and a decrease in overall tissue damage in the CNS compared with isotype control-treated mice. Ccr5 is crucial in directing T cells toward the LGTV-infected brain, as well as in suppressing neutrophil-mediated inflammation within the CNS.
Copyright © 2016 by The American Association of Immunologists, Inc.
Figures
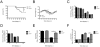
Similar articles
-
Type I interferon protects mice from fatal neurotropic infection with Langat virus by systemic and local antiviral responses.J Virol. 2014 Nov;88(21):12202-12. doi: 10.1128/JVI.01215-14. Epub 2014 Aug 13. J Virol. 2014. PMID: 25122777 Free PMC article.
-
Stable and Highly Immunogenic MicroRNA-Targeted Single-Dose Live Attenuated Vaccine Candidate against Tick-Borne Encephalitis Constructed Using Genetic Backbone of Langat Virus.mBio. 2019 Apr 23;10(2):e02904-18. doi: 10.1128/mBio.02904-18. mBio. 2019. PMID: 31015334 Free PMC article.
-
Cross-reactive antibodies against Langat virus protect mice from lethal tick-borne encephalitis virus infection.Front Immunol. 2023 Feb 28;14:1134371. doi: 10.3389/fimmu.2023.1134371. eCollection 2023. Front Immunol. 2023. PMID: 36926332 Free PMC article.
-
Cell-Mediated Immune Responses and Immunopathogenesis of Human Tick-Borne Encephalitis Virus-Infection.Front Immunol. 2018 Sep 26;9:2174. doi: 10.3389/fimmu.2018.02174. eCollection 2018. Front Immunol. 2018. PMID: 30319632 Free PMC article. Review.
-
Host immunogenetics in tick-borne encephalitis virus infection-The CCR5 crossroad.Ticks Tick Borne Dis. 2019 Jun;10(4):729-741. doi: 10.1016/j.ttbdis.2019.03.005. Epub 2019 Mar 11. Ticks Tick Borne Dis. 2019. PMID: 30879988 Review.
Cited by
-
Neuroinvasive Flavivirus Pathogenesis Is Restricted by Host Genetic Factors in Collaborative Cross Mice, Independently of Oas1b.J Virol. 2023 Jul 27;97(7):e0071523. doi: 10.1128/jvi.00715-23. Epub 2023 Jun 13. J Virol. 2023. PMID: 37310228 Free PMC article.
-
Tick-borne encephalitis affects sleep-wake behavior and locomotion in infant rats.Cell Biosci. 2022 Aug 2;12(1):121. doi: 10.1186/s13578-022-00859-7. Cell Biosci. 2022. PMID: 35918749 Free PMC article.
-
Chemokine receptor CCR5 correlates with functional CD8+ T cells in SIV-infected macaques and the potential effects of maraviroc on T-cell activation.FASEB J. 2019 Aug;33(8):8905-8912. doi: 10.1096/fj.201802703R. Epub 2019 Apr 29. FASEB J. 2019. PMID: 31034775 Free PMC article.
-
The CCR5 antagonist maraviroc exerts limited neuroprotection without improving neurofunctional outcome in experimental pneumococcal meningitis.Sci Rep. 2022 Jul 28;12(1):12945. doi: 10.1038/s41598-022-17282-0. Sci Rep. 2022. PMID: 35902720 Free PMC article.
-
An Overview of Animal Models for Arthropod-Borne Viruses.Comp Med. 2017 Jun 1;67(3):232-241. Comp Med. 2017. PMID: 28662752 Free PMC article. Review.
References
-
- Donoso-Mantke O, K.L.S., Ruzek D. InTechOpen. 1st ed. 2011. Tick-Borne Encephalitis Virus: A General Overview.
-
- Gritsun TS, Lashkevich VA, Gould EA. Tick-borne encephalitis. Antiviral research. 2003;57:129–146. - PubMed
-
- Suss J. Tick-borne encephalitis in Europe and beyond--the epidemiological situation as of 2007. Euro surveillance : bulletin Europeen sur les maladies transmissibles = European communicable disease bulletin. 2008;13 - PubMed
-
- Ruzek D, Dobler G, Donoso Mantke O. Tick-borne encephalitis: pathogenesis and clinical implications. Travel medicine and infectious disease. 2010;8:223–232. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

