Regulation of KAT6 Acetyltransferases and Their Roles in Cell Cycle Progression, Stem Cell Maintenance, and Human Disease
- PMID: 27185879
- PMCID: PMC4936061
- DOI: 10.1128/MCB.00055-16
Regulation of KAT6 Acetyltransferases and Their Roles in Cell Cycle Progression, Stem Cell Maintenance, and Human Disease
Abstract
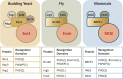
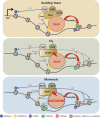
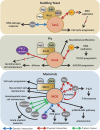
The lysine acetyltransferase 6 (KAT6) histone acetyltransferase (HAT) complexes are highly conserved from yeast to higher organisms. They acetylate histone H3 and other nonhistone substrates and are involved in cell cycle regulation and stem cell maintenance. In addition, the human KAT6 HATs are recurrently mutated in leukemia and solid tumors. Therefore, it is important to understand the mechanisms underlying the regulation of KAT6 HATs and their roles in cell cycle progression. In this minireview, we summarize the identification and analysis of the KAT6 complexes and discuss the regulatory mechanisms governing their enzymatic activities and substrate specificities. We further focus on the roles of KAT6 HATs in regulating cell proliferation and stem cell maintenance and review recent insights that aid in understanding their involvement in human diseases.
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures
Similar articles
-
The key roles of the lysine acetyltransferases KAT6A and KAT6B in physiology and pathology.Drug Resist Updat. 2020 Dec;53:100729. doi: 10.1016/j.drup.2020.100729. Epub 2020 Oct 7. Drug Resist Updat. 2020. PMID: 33130515 Review.
-
Aberrant forms of histone acetyltransferases in human disease.Subcell Biochem. 2007;41:233-62. Subcell Biochem. 2007. PMID: 17484131 Review.
-
Lysine Acetyltransferase 6 Complexes in Neurodevelopmental Disorders and Different Types of Cancer.Results Probl Cell Differ. 2025;75:391-410. doi: 10.1007/978-3-031-91459-1_14. Results Probl Cell Differ. 2025. PMID: 40593218 Review.
-
Cancers with wrong HATs: the impact of acetylation.Brief Funct Genomics. 2013 May;12(3):231-43. doi: 10.1093/bfgp/els065. Epub 2013 Jan 15. Brief Funct Genomics. 2013. PMID: 23325510 Review.
-
Histone H3 specific acetyltransferases are essential for cell cycle progression.Genes Dev. 2001 Dec 1;15(23):3144-54. doi: 10.1101/gad.931401. Genes Dev. 2001. PMID: 11731478 Free PMC article.
Cited by
-
Limiting PCNA-unloading at the G1/S transition.Cell Cycle. 2016 Nov 16;15(22):3001-3002. doi: 10.1080/15384101.2016.1214036. Epub 2016 Jul 27. Cell Cycle. 2016. PMID: 27463534 Free PMC article. No abstract available.
-
Impaired Regulation of Histone Methylation and Acetylation Underlies Specific Neurodevelopmental Disorders.Front Genet. 2021 Jan 8;11:613098. doi: 10.3389/fgene.2020.613098. eCollection 2020. Front Genet. 2021. PMID: 33488679 Free PMC article. Review.
-
HBO1, a MYSTerious KAT and its links to cancer.Biochim Biophys Acta Gene Regul Mech. 2024 Sep;1867(3):195045. doi: 10.1016/j.bbagrm.2024.195045. Epub 2024 Jun 6. Biochim Biophys Acta Gene Regul Mech. 2024. PMID: 38851533 Review.
-
KAT6A chimeras form a self-reinforcing epigenetic module with NURF and MLL/COMPASS to sustain AML.Genome Biol. 2025 Aug 19;26(1):253. doi: 10.1186/s13059-025-03743-y. Genome Biol. 2025. PMID: 40830799 Free PMC article.
-
Metagenomic characterization of lysine acetyltransferases in human cancer and their association with clinicopathologic features.Cancer Sci. 2020 May;111(5):1829-1839. doi: 10.1111/cas.14385. Epub 2020 Apr 24. Cancer Sci. 2020. PMID: 32162442 Free PMC article.
References
-
- Steunou A-L, Rossetto D, Côté J. 2014. Regulating chromatin by histone acetylation, p 147–212. In Workman JL, Abmayr SM (ed), Fundamentals of chromatin, 1st ed Springer, New York, NY. doi:10.1007/978-1-4614-8624-4. - DOI
-
- Borrow J, Stanton VP Jr, Andresen JM, Becher R, Behm FG, Chaganti RS, Civin CI, Disteche C, Dube I, Frischauf AM, Horsman D, Mitelman F, Volinia S, Watmore AE, Housman DE. 1996. The translocation t(8;16)(p11;p13) of acute myeloid leukaemia fuses a putative acetyltransferase to the CREB-binding protein. Nat Genet 14:33–41. doi:10.1038/ng0996-33. - DOI - PubMed
-
- Arboleda VA, Lee H, Dorrani N, Zadeh N, Willis M, Macmurdo CF, Manning MA, Kwan A, Hudgins L, Barthelemy F, Miceli MC, Quintero-Rivera F, Kantarci S, Strom SP, Deignan JL, Center UCG, Grody WW, Vilain E, Nelson SF. 2015. De novo nonsense mutations in KAT6A, a lysine acetyl-transferase gene, cause a syndrome including microcephaly and global developmental delay. Am J Hum Genet 96:498–506. doi:10.1016/j.ajhg.2015.01.017. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
