Water Determines the Structure and Dynamics of Proteins
- PMID: 27186992
- PMCID: PMC7116073
- DOI: 10.1021/acs.chemrev.5b00664
Water Determines the Structure and Dynamics of Proteins
Abstract
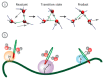
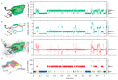
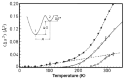
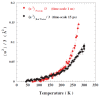
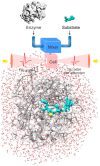
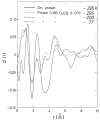
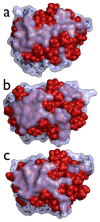
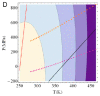
Water is an essential participant in the stability, structure, dynamics, and function of proteins and other biomolecules. Thermodynamically, changes in the aqueous environment affect the stability of biomolecules. Structurally, water participates chemically in the catalytic function of proteins and nucleic acids and physically in the collapse of the protein chain during folding through hydrophobic collapse and mediates binding through the hydrogen bond in complex formation. Water is a partner that slaves the dynamics of proteins, and water interaction with proteins affect their dynamics. Here we provide a review of the experimental and computational advances over the past decade in understanding the role of water in the dynamics, structure, and function of proteins. We focus on the combination of X-ray and neutron crystallography, NMR, terahertz spectroscopy, mass spectroscopy, thermodynamics, and computer simulations to reveal how water assist proteins in their function. The recent advances in computer simulations and the enhanced sensitivity of experimental tools promise major advances in the understanding of protein dynamics, and water surely will be a protagonist.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

References
-
- Kauzmann W. Some Factors in The Interpretation of Protein Denaturation. Adv Protein Chem. 1959;14:1–63. - PubMed
-
- Anfinsen CB. Principles That Govern The Folding of Protein Chains. Science. 1973;181(4096):223–230. - PubMed
-
- Hartl FU, Bracher A, Hayer-Hartl M. Molecular Chaperones in Protein Folding and Proteostasis. Nature. 2011;475(7356):324–332. - PubMed
-
- Kim YE, Hipp MS, Bracher A, Hayer-Hartl M, Hartl FU. Molecular Chaperone Functions in Protein Folding and Proteostasis. Annu Rev Biochem. 2013;82:323–355. - PubMed
-
- Makhatadze GI, Privalov PL. Contribution of Hydration To Protein-Folding Thermodynamics 0.1. The Enthalpy of Hydration. J Mol Biol. 1993;232(2):639–659. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

