Blood coagulation factor XII drives adaptive immunity during neuroinflammation via CD87-mediated modulation of dendritic cells
- PMID: 27188843
- PMCID: PMC4873982
- DOI: 10.1038/ncomms11626
Blood coagulation factor XII drives adaptive immunity during neuroinflammation via CD87-mediated modulation of dendritic cells
Abstract
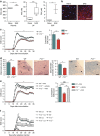
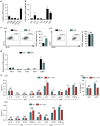
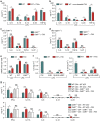
Aberrant immune responses represent the underlying cause of central nervous system (CNS) autoimmunity, including multiple sclerosis (MS). Recent evidence implicated the crosstalk between coagulation and immunity in CNS autoimmunity. Here we identify coagulation factor XII (FXII), the initiator of the intrinsic coagulation cascade and the kallikrein-kinin system, as a specific immune cell modulator. High levels of FXII activity are present in the plasma of MS patients during relapse. Deficiency or pharmacologic blockade of FXII renders mice less susceptible to experimental autoimmune encephalomyelitis (a model of MS) and is accompanied by reduced numbers of interleukin-17A-producing T cells. Immune activation by FXII is mediated by dendritic cells in a CD87-dependent manner and involves alterations in intracellular cyclic AMP formation. Our study demonstrates that a member of the plasmatic coagulation cascade is a key mediator of autoimmunity. FXII inhibition may provide a strategy to combat MS and other immune-related disorders.
Conflict of interest statement
M.W.N. is an employee of CSL Behring GmbH and owns employee shares in CSL Limited. C.P. is an employee of CSL Limited and owns employee shares in CSL Limited. K.G., M.W.N., C.K. and S.G.M. hold a patent to treat neuroinflammatory disorders with FXIIa inhibitors. The remaining authors declare no competing financial interests.
Figures




Similar articles
-
Coagulation factor XII induces pro-inflammatory cytokine responses in macrophages and promotes atherosclerosis in mice.Thromb Haemost. 2017 Jan 5;117(1):176-187. doi: 10.1160/TH16-06-0466. Epub 2016 Oct 27. Thromb Haemost. 2017. PMID: 27786338
-
Plasma kallikrein modulates immune cell trafficking during neuroinflammation via PAR2 and bradykinin release.Proc Natl Acad Sci U S A. 2019 Jan 2;116(1):271-276. doi: 10.1073/pnas.1810020116. Epub 2018 Dec 17. Proc Natl Acad Sci U S A. 2019. PMID: 30559188 Free PMC article.
-
Targeting the GM-CSF receptor for the treatment of CNS autoimmunity.J Autoimmun. 2017 Nov;84:1-11. doi: 10.1016/j.jaut.2017.06.005. Epub 2017 Jun 20. J Autoimmun. 2017. PMID: 28641926 Free PMC article.
-
G Protein-Coupled Kinin Receptors and Immunity Against Pathogens.Adv Immunol. 2017;136:29-84. doi: 10.1016/bs.ai.2017.05.007. Epub 2017 Aug 23. Adv Immunol. 2017. PMID: 28950949 Review.
-
Pharmacological regulation of factor XII activation may be a new target to control pathological coagulation.Biochem Pharmacol. 2008 Mar 1;75(5):1007-13. doi: 10.1016/j.bcp.2007.10.003. Epub 2007 Oct 7. Biochem Pharmacol. 2008. PMID: 17996217 Review.
Cited by
-
Neuroprotective Strategies in Aneurysmal Subarachnoid Hemorrhage (aSAH).Int J Mol Sci. 2021 May 21;22(11):5442. doi: 10.3390/ijms22115442. Int J Mol Sci. 2021. PMID: 34064048 Free PMC article. Review.
-
Triazol-1-yl Benzamides Promote Anticoagulant Activity via Inhibition of Factor XIIa.Cardiovasc Hematol Agents Med Chem. 2023;21(2):108-119. doi: 10.2174/1871525721666221031141323. Cardiovasc Hematol Agents Med Chem. 2023. PMID: 36321236 Free PMC article.
-
The contact system in liver injury.Semin Immunopathol. 2021 Aug;43(4):507-517. doi: 10.1007/s00281-021-00876-7. Epub 2021 Jun 14. Semin Immunopathol. 2021. PMID: 34125270 Free PMC article. Review.
-
Dysregulated haemostasis in thrombo-inflammatory disease.Clin Sci (Lond). 2022 Dec 22;136(24):1809-1829. doi: 10.1042/CS20220208. Clin Sci (Lond). 2022. PMID: 36524413 Free PMC article. Review.
-
Activation of the Coagulation Cascade as a Universal Danger Sign.Curr Issues Mol Biol. 2025 Feb 9;47(2):108. doi: 10.3390/cimb47020108. Curr Issues Mol Biol. 2025. PMID: 39996829 Free PMC article. Review.
References
-
- Bhat R. & Steinman L. Innate and adaptive autoimmunity directed to the central nervous system. Neuron 64, 123–132 (2009). - PubMed
-
- Steinman L. A brief history of T(H)17, the first major revision in the T(H)1/T(H)2 hypothesis of T cell-mediated tissue damage. Nat. Med. 13, 139–145 (2007). - PubMed
-
- Korn T., Bettelli E., Oukka M. & IL-17 Kuchroo VK. and Th17 Cells. Annu. Rev. Immunol. 27, 485–517 (2009). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

