Risk of long-term anticoagulation under sustained severe arterial hypertension: A translational study comparing warfarin and the new oral anticoagulant apixaban
- PMID: 27189904
- PMCID: PMC5363464
- DOI: 10.1177/0271678X16642443
Risk of long-term anticoagulation under sustained severe arterial hypertension: A translational study comparing warfarin and the new oral anticoagulant apixaban
Abstract
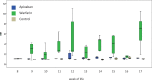
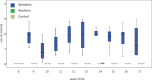
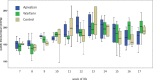
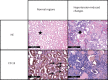
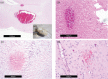
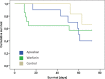
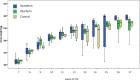
New oral anticoagulants for the prevention of stroke and systemic embolism in patients with atrial fibrillation have recently been introduced. In this translational study, we explored the risk of long-term anticoagulation on intracerebral hemorrhage under sustained severe arterial hypertension. We initiated anticoagulation with warfarin or apixaban in spontaneously hypertensive rats prone to develop severe hypertension and subsequent intracerebral bleeding complications. A non-anticoagulated group served as control. During an 11-week-study period, blood pressure, anticoagulation parameters, and clinical status were determined regularly. The incidence of histopathologically proven intracerebral hemorrhage was defined as the primary endpoint. Both warfarin and apixaban anticoagulation was fairly stable during the study period, and all rats developed severe hypertension. Intracerebral hemorrhage was determined in 29% (4/14) of warfarin rats and in 10% (1/10) of apixaban rats. Controls did not show cerebral bleeding complications (chi-square not significant). Mortality rate at study termination was 33% (2/6) in controls, 43% (6/14) in the warfarin group, and 60% (6/10) in the apixaban group. Animals died from extracerebral complications in most cases. Our study describes an experimental intracerebral hemorrhage model in the context of sustained hypertension and long-term anticoagulation. Extracerebral bleeding complications occurred more often in warfarin-treated animals compared with apixaban and control rats.
Keywords: Antithrombotics; animal model; brain ischemia; hypertension; intracerebral hemorrhage.
Figures
Similar articles
-
Novel oral anticoagulants for stroke prevention in atrial fibrillation: focus on apixaban.Adv Ther. 2012 Jun;29(6):491-507. doi: 10.1007/s12325-012-0026-8. Epub 2012 Jun 7. Adv Ther. 2012. PMID: 22684583 Review.
-
Apixaban versus Warfarin for the Prevention of Periprocedural Cerebral Thromboembolism in Atrial Fibrillation Ablation: Multicenter Prospective Randomized Study.J Cardiovasc Electrophysiol. 2016 May;27(5):549-54. doi: 10.1111/jce.12928. Epub 2016 Feb 12. J Cardiovasc Electrophysiol. 2016. PMID: 26766541 Clinical Trial.
-
Apixaban versus warfarin in patients with atrial fibrillation according to prior warfarin use: results from the Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation trial.Am Heart J. 2013 Sep;166(3):549-58. doi: 10.1016/j.ahj.2013.05.016. Epub 2013 Jul 25. Am Heart J. 2013. PMID: 24016506 Clinical Trial.
-
Apixaban for stroke prevention in atrial fibrillation: a review of the clinical trial evidence.Hosp Pract (1995). 2011 Oct;39(4):7-16. doi: 10.3810/hp.2011.10.918. Hosp Pract (1995). 2011. PMID: 22056819 Review.
-
Non-major bleeding with apixaban versus warfarin in patients with atrial fibrillation.Heart. 2017 Apr;103(8):623-628. doi: 10.1136/heartjnl-2016-309901. Epub 2016 Oct 24. Heart. 2017. PMID: 27798052 Free PMC article. Clinical Trial.
References
-
- January CT, Wann LS, Alpert JS, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: A report of the American College of Cardiology/American Heart Association task force on practice guidelines and the heart rhythm society. Circulation 2014; 130: e199–267. - PMC - PubMed
-
- Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 2009; 361: 1139–1151. - PubMed
-
- Granger CB, Alexander JH, McMurray JJ, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med 2011; 365: 981–992. - PubMed
-
- Patel MR, Mahaffey KW, Garg J, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 2011; 365: 883–891. - PubMed
-
- Ansell J. New oral anticoagulants should not be used as first-line agents to prevent thromboembolism in patients with atrial fibrillation. Circulation 2012; 125: 165–170. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

