Aging-related alterations in eNOS and nNOS responsiveness and smooth muscle reactivity of murine basilar arteries are modulated by apocynin and phosphorylation of myosin phosphatase targeting subunit-1
- PMID: 27193035
- PMCID: PMC5363478
- DOI: 10.1177/0271678X16649402
Aging-related alterations in eNOS and nNOS responsiveness and smooth muscle reactivity of murine basilar arteries are modulated by apocynin and phosphorylation of myosin phosphatase targeting subunit-1
Abstract
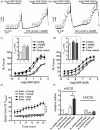
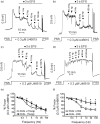
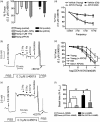
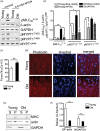
Aging causes major alterations of all components of the neurovascular unit and compromises brain blood supply. Here, we tested how aging affects vascular reactivity in basilar arteries from young (<10 weeks; y-BA), old (>22 months; o-BA) and old (>22 months) heterozygous MYPT1-T-696A/+ knock-in mice. In isometrically mounted o-BA, media thickness was increased by ∼10% while the passive length tension relations were not altered. Endothelial denudation or pan-NOS inhibition (100 µmol/L L-NAME) increased the basal tone by 11% in y-BA and 23% in o-BA, while inhibition of nNOS (1 µmol/L L-NPA) induced ∼10% increase in both ages. eNOS expression was ∼2-fold higher in o-BA. In o-BA, U46619-induced force was augmented (pEC50 ∼6.9 vs. pEC50 ∼6.5) while responsiveness to DEA-NONOate, electrical field stimulation or nicotine was decreased. Basal phosphorylation of MLC20-S19 and MYPT1-T-853 was higher in o-BA and was reversed by apocynin. Furthermore, permeabilized o-BA showed enhanced Ca2+-sensitivity. Old T-696A/+ BA displayed a reduced phosphorylation of MYPT1-T696 and MLC20, a lower basal tone in response to L-NAME and a reduced eNOS expression. The results indicate that the vascular hypercontractility found in o-BA is mediated by inhibition of MLCP and is partially compensated by an upregulation of endothelial NO release.
Keywords: Aging; MYPT1-T696 gene mutation; actin dynamics; basilar artery; neurovascular uncoupling; vascular hypercontractility.
Figures

Similar articles
-
Alanine mutation of the targeting subunit of the myosin phosphatase, MYPT1 at threonine 696 reduces cGMP responsiveness of mouse femoral arteries.Eur J Pharmacol. 2025 Jan 5;986:177133. doi: 10.1016/j.ejphar.2024.177133. Epub 2024 Nov 17. Eur J Pharmacol. 2025. PMID: 39551336
-
The involvement of phosphorylation of myosin phosphatase targeting subunit 1 (MYPT1) and MYPT1 isoform expression in NO/cGMP mediated differential vasoregulation of cerebral arteries compared to systemic arteries.Acta Physiol (Oxf). 2018 Sep;224(1):e13079. doi: 10.1111/apha.13079. Epub 2018 May 22. Acta Physiol (Oxf). 2018. PMID: 29694711
-
Dual thick and thin filament linked regulation of stretch- and L-NAME-induced tone in young and senescent murine basilar artery.Front Physiol. 2023 Mar 28;14:1099278. doi: 10.3389/fphys.2023.1099278. eCollection 2023. Front Physiol. 2023. PMID: 37057180 Free PMC article.
-
Regulation of Smooth Muscle Myosin Light Chain Phosphatase by Multisite Phosphorylation of the Myosin Targeting Subunit, MYPT1.Cardiovasc Hematol Disord Drug Targets. 2018;18(1):4-13. doi: 10.2174/1871529X18666180326120638. Cardiovasc Hematol Disord Drug Targets. 2018. PMID: 29577868 Review.
-
Regulation of Myosin Light-Chain Phosphatase Activity to Generate Airway Smooth Muscle Hypercontractility.Front Physiol. 2020 Jun 26;11:701. doi: 10.3389/fphys.2020.00701. eCollection 2020. Front Physiol. 2020. PMID: 32676037 Free PMC article. Review.
Cited by
-
Caldesmon ablation in mice causes umbilical herniation and alters contractility of fetal urinary bladder smooth muscle.J Gen Physiol. 2021 Jul 5;153(7):e202012776. doi: 10.1085/jgp.202012776. Epub 2021 Jun 11. J Gen Physiol. 2021. PMID: 34115104 Free PMC article.
-
Augmented contractility of murine femoral arteries in a streptozotocin diabetes model is related to increased phosphorylation of MYPT1.Physiol Rep. 2019 Feb;7(3):e13975. doi: 10.14814/phy2.13975. Physiol Rep. 2019. PMID: 30740930 Free PMC article.
-
Emerging Roles of Endothelial Nitric Oxide in Preservation of Cognitive Health.Stroke. 2023 Mar;54(3):686-696. doi: 10.1161/STROKEAHA.122.041444. Epub 2023 Feb 27. Stroke. 2023. PMID: 36848426 Free PMC article. Review.
-
Indoxyl sulfate enhances endothelin-1-induced contraction via impairment of NO/cGMP signaling in rat aorta.Pflugers Arch. 2021 Aug;473(8):1247-1259. doi: 10.1007/s00424-021-02581-8. Epub 2021 May 22. Pflugers Arch. 2021. PMID: 34021781
-
Interplay of myosin phosphatase and protein phosphatase-2A in the regulation of endothelial nitric-oxide synthase phosphorylation and nitric oxide production.Sci Rep. 2017 Mar 16;7:44698. doi: 10.1038/srep44698. Sci Rep. 2017. PMID: 28300193 Free PMC article.
References
-
- Park L, Anrather J, Girouard H, et al. Nox2-derived reactive oxygen species mediate neurovascular dysregulation in the aging mouse brain. J Cereb Blood Flow Metab 2007; 27: 1908–1918. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

