Preserved Activity of CD20-Specific Chimeric Antigen Receptor-Expressing T Cells in the Presence of Rituximab
- PMID: 27197068
- PMCID: PMC4891234
- DOI: 10.1158/2326-6066.CIR-15-0276
Preserved Activity of CD20-Specific Chimeric Antigen Receptor-Expressing T Cells in the Presence of Rituximab
Abstract
CD20 is an attractive immunotherapy target for B-cell non-Hodgkin lymphomas, and adoptive transfer of T cells genetically modified to express a chimeric antigen receptor (CAR) targeting CD20 is a promising strategy. A theoretical limitation is that residual serum rituximab might block CAR binding to CD20 and thereby impede T cell-mediated anti-lymphoma responses. The activity of CD20 CAR-modified T cells in the presence of various concentrations of rituximab was tested in vitro and in vivo CAR-binding sites on CD20(+) tumor cells were blocked by rituximab in a dose-dependent fashion, although at 37°C blockade was incomplete at concentrations up to 200 μg/mL. T cells with CD20 CARs also exhibited modest dose-dependent reductions in cytokine secretion and cytotoxicity, but not proliferation, against lymphoma cell lines. At rituximab concentrations of 100 μg/mL, CAR T cells retained ≥50% of baseline activity against targets with high CD20 expression, but were more strongly inhibited when target cells expressed low CD20. In a murine xenograft model using a rituximab-refractory lymphoma cell line, rituximab did not impair CAR T-cell activity, and tumors were eradicated in >85% of mice. Clinical residual rituximab serum concentrations were measured in 103 lymphoma patients after rituximab therapy, with the median level found to be only 38 μg/mL (interquartile range, 19-72 μg/mL). Thus, despite modest functional impairment in vitro, the in vivo activity of CD20-targeted CAR T cells remains intact at clinically relevant levels of rituximab, making use of these T cells clinically feasible. Cancer Immunol Res; 4(6); 509-19. ©2016 AACR
©2016 American Association for Cancer Research.
Figures
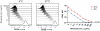

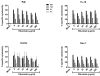
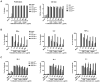

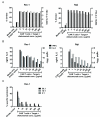
Comment in
-
Tales of Antigen Evasion from CAR Therapy.Cancer Immunol Res. 2016 Jun;4(6):473. doi: 10.1158/2326-6066.CIR-16-0089. Cancer Immunol Res. 2016. PMID: 27252092
References
-
- Kochenderfer JN, Dudley ME, Kassim SH, Somerville RP, Carpenter RO, Stetler-Stevenson M, et al. Chemotherapy-refractory diffuse large B-cell lymphoma and indolent B-cell malignancies can be effectively treated with autologous T cells expressing an anti-CD19 chimeric antigen receptor. J Clin Oncol. 2015;33(6):540–9. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

