Real-world results of ibrutinib in patients with relapsed or refractory chronic lymphocytic leukemia: data from 95 consecutive patients treated in a compassionate use program. A study from the Swedish Chronic Lymphocytic Leukemia Group
- PMID: 27198718
- PMCID: PMC5479603
- DOI: 10.3324/haematol.2016.144576
Real-world results of ibrutinib in patients with relapsed or refractory chronic lymphocytic leukemia: data from 95 consecutive patients treated in a compassionate use program. A study from the Swedish Chronic Lymphocytic Leukemia Group
Abstract
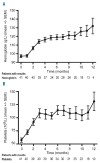
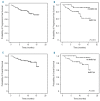
Ibrutinib, a Bruton's tyrosine kinase inhibitor is approved for relapsed/refractory and del(17p)/TP53 mutated chronic lymphocytic leukemia. Discrepancies between clinical trials and routine health-care are commonly observed in oncology. Herein we report real-world results for 95 poor prognosis Swedish patients treated with ibrutinib in a compassionate use program. Ninety-five consecutive patients (93 chronic lymphocytic leukemia, 2 small lymphocytic leukemia) were included in the study between May 2014 and May 2015. The median age was 69 years. 63% had del(17p)/TP53 mutation, 65% had Rai stage III/IV, 28% had lymphadenopathy ≥10cm. Patients received ibrutinib 420 mg once daily until progression. At a median follow-up of 10.2 months, the overall response rate was 84% (consistent among subgroups) and 77% remained progression-free. Progression-free survival and overall survival were significantly shorter in patients with del(17p)/TP53 mutation (P=0.017 and P=0.027, log-rank test); no other factor was significant in Cox proportional regression hazards model. Ibrutinib was well tolerated. Hematomas occurred in 46% of patients without any major bleeding. Seven patients had Richter's transformation. This real-world analysis on consecutive chronic lymphocytic leukemia patients from a well-defined geographical region shows the efficacy and safety of ibrutinib to be similar to that of pivotal trials. Yet, del(17p)/TP53 mutation remains a therapeutic challenge. Since not more than half of our patients would have qualified for the pivotal ibrutinib trial (RESONATE), our study emphasizes that real-world results should be carefully considered in future with regards to new agents and new indications in chronic lymphocytic leukemia.
Copyright© Ferrata Storti Foundation.
Figures
References
-
- Hallek M, Fischer K, Fingerle-Rowson G, et al. Addition of rituximab to fludarabine and cyclophosphamide in patients with chronic lymphocytic leukaemia: a randomised, open-label, phase 3 trial. Lancet. 2010;376(9747):1164–1174. - PubMed
-
- Zenz T, Eichhorst B, Busch R, et al. TP53 mutation and survival in chronic lymphocytic leukemia. J Clin Oncol. 2010;28(29): 4473–4479. - PubMed
-
- Eketorp Sylvan S, Hansson L, Karlsson C, et al. Outcomes of patients with fludarabine-refractory chronic lymphocytic leukemia: a population-based study from a well-defined geographic region. Leuk Lymphoma. 2014;55(8):1774–1780. - PubMed
-
- Tam CS, O’Brien S, Lerner S, et al. The natural history of fludarabine-refractory chronic lymphocytic leukemia patients who fail alemtuzumab or have bulky lymphadenopathy. Leuk Lymphoma. 2007;48(10):1931–1939. - PubMed
-
- Pan Z, Scheerens H, Li SJ, et al. Discovery of selective irreversible inhibitors for Bruton’s tyrosine kinase. ChemMedChem. 2007;2(1):58–61. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

