The Role of Slr0151, a Tetratricopeptide Repeat Protein from Synechocystis sp. PCC 6803, during Photosystem II Assembly and Repair
- PMID: 27200072
- PMCID: PMC4853703
- DOI: 10.3389/fpls.2016.00605
The Role of Slr0151, a Tetratricopeptide Repeat Protein from Synechocystis sp. PCC 6803, during Photosystem II Assembly and Repair
Abstract
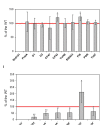
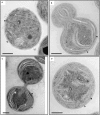
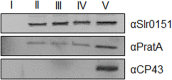
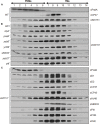
The assembly and repair of photosystem II (PSII) is facilitated by a variety of assembly factors. Among those, the tetratricopeptide repeat (TPR) protein Slr0151 from Synechocystis sp. PCC 6803 (hereafter Synechocystis) has previously been assigned a repair function under high light conditions (Yang et al., 2014). Here, we show that inactivation of slr0151 affects thylakoid membrane ultrastructure even under normal light conditions. Moreover, the level and localization of Slr0151 are affected in a variety of PSII-related mutants. In particular, the data suggest a close functional relationship between Slr0151 and Sll0933, which interacts with Ycf48 during PSII assembly and is homologous to PAM68 in Arabidopsis thaliana. Immunofluorescence analysis revealed a punctate distribution of Slr0151 within several different membrane types in Synechocystis cells.
Keywords: Synechocystis; TPR protein; biogenesis center; photosystem II; thylakoid membrane.
Figures
Similar articles
-
Slr0151 in Synechocystis sp. PCC 6803 is required for efficient repair of photosystem II under high-light condition.J Integr Plant Biol. 2014 Dec;56(12):1136-50. doi: 10.1111/jipb.12275. Epub 2014 Sep 25. J Integr Plant Biol. 2014. PMID: 25146729
-
The cyanobacterial homologue of HCF136/YCF48 is a component of an early photosystem II assembly complex and is important for both the efficient assembly and repair of photosystem II in Synechocystis sp. PCC 6803.J Biol Chem. 2008 Aug 15;283(33):22390-9. doi: 10.1074/jbc.M801917200. Epub 2008 Jun 12. J Biol Chem. 2008. PMID: 18550538
-
Characterization of a Synechocystis double mutant lacking the photosystem II assembly factors YCF48 and Sll0933.Planta. 2013 Feb;237(2):471-80. doi: 10.1007/s00425-012-1720-0. Epub 2012 Jul 31. Planta. 2013. PMID: 22847023
-
Identification and Roles of Photosystem II Assembly, Stability, and Repair Factors in Arabidopsis.Front Plant Sci. 2016 Feb 16;7:168. doi: 10.3389/fpls.2016.00168. eCollection 2016. Front Plant Sci. 2016. PMID: 26909098 Free PMC article. Review.
-
Photosystem II repair in plant chloroplasts--Regulation, assisting proteins and shared components with photosystem II biogenesis.Biochim Biophys Acta. 2015 Sep;1847(9):900-9. doi: 10.1016/j.bbabio.2015.01.006. Epub 2015 Jan 21. Biochim Biophys Acta. 2015. PMID: 25615587 Review.
Cited by
-
Elucidating the origins of phycocyanobilin biosynthesis and phycobiliproteins.Proc Natl Acad Sci U S A. 2023 Apr 25;120(17):e2300770120. doi: 10.1073/pnas.2300770120. Epub 2023 Apr 18. Proc Natl Acad Sci U S A. 2023. PMID: 37071675 Free PMC article.
-
Patterning of the Autotrophic, Mixotrophic, and Heterotrophic Proteomes of Oxygen-Evolving Cyanobacterium Synechocystis sp. PCC 6803.Front Microbiol. 2022 May 25;13:891895. doi: 10.3389/fmicb.2022.891895. eCollection 2022. Front Microbiol. 2022. PMID: 35694301 Free PMC article.
-
Mass spectrometry analysis of the photosystem II assembly factor Psb27 revealed variations in its lipid modification.Photosynth Res. 2022 Jun;152(3):305-316. doi: 10.1007/s11120-021-00891-7. Epub 2021 Dec 15. Photosynth Res. 2022. PMID: 34910272 Free PMC article.
-
Thylakoid Membrane Architecture in Synechocystis Depends on CurT, a Homolog of the Granal CURVATURE THYLAKOID1 Proteins.Plant Cell. 2016 Sep;28(9):2238-2260. doi: 10.1105/tpc.16.00491. Epub 2016 Aug 19. Plant Cell. 2016. PMID: 27543090 Free PMC article.
-
Recent advances in understanding photosynthesis.F1000Res. 2016 Dec 21;5:2890. doi: 10.12688/f1000research.9744.1. eCollection 2016. F1000Res. 2016. PMID: 28105322 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

