Insulin decreases atherosclerosis by inducing endothelin receptor B expression
- PMID: 27200419
- PMCID: PMC4869734
- DOI: 10.1172/jci.insight.86574
Insulin decreases atherosclerosis by inducing endothelin receptor B expression
Abstract
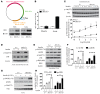
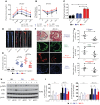
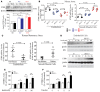
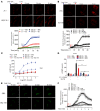
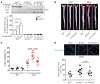
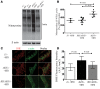
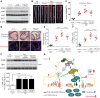
Endothelial cell (EC) insulin resistance and dysfunction, caused by diabetes, accelerates atherosclerosis. It is unknown whether specifically enhancing EC-targeted insulin action can decrease atherosclerosis in diabetes. Accordingly, overexpressing insulin receptor substrate-1 (IRS1) in the endothelia of Apoe-/- mice (Irs1/Apoe-/-) increased insulin signaling and function in the aorta. Atherosclerosis was significantly reduced in Irs1/ApoE-/- mice on diet-induced hyperinsulinemia and hyperglycemia. The mechanism of insulin's enhanced antiatherogenic actions in EC was related to remarkable induction of NO action, which increases endothelin receptor B (EDNRB) expression and intracellular [Ca2+]. Using the mice with knockin mutation of eNOS, which had Ser1176 mutated to alanine (AKI), deleting the only known mechanism for insulin to activate eNOS/NO pathway, we observed that IRS1 overexpression in the endothelia of Aki/ApoE-/- mice significantly decreased atherosclerosis. Interestingly, endothelial EDNRB expression was selectively reduced in intima of arteries from diabetic patients and rodents. However, endothelial EDNRB expression was upregulated by insulin via P13K/Akt pathway. Finally EDNRB deletion in EC of Ldlr-/- and Irs1/Ldlr-/- mice decreased NO production and accelerated atherosclerosis, compared with Ldlr-/- mice. Accelerated atherosclerosis in diabetes may be reduced by improving insulin signaling selectively via IRS1/Akt in the EC by inducing EDNRB expression and NO production.
Figures


References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

