Biophysical Network Modelling of the dLGN Circuit: Different Effects of Triadic and Axonal Inhibition on Visual Responses of Relay Cells
- PMID: 27203421
- PMCID: PMC4874694
- DOI: 10.1371/journal.pcbi.1004929
Biophysical Network Modelling of the dLGN Circuit: Different Effects of Triadic and Axonal Inhibition on Visual Responses of Relay Cells
Abstract
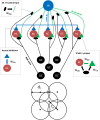
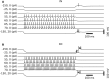
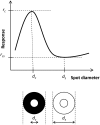
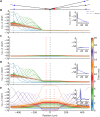
Despite its prominent placement between the retina and primary visual cortex in the early visual pathway, the role of the dorsal lateral geniculate nucleus (dLGN) in molding and regulating the visual signals entering the brain is still poorly understood. A striking feature of the dLGN circuit is that relay cells (RCs) and interneurons (INs) form so-called triadic synapses, where an IN dendritic terminal can be simultaneously postsynaptic to a retinal ganglion cell (GC) input and presynaptic to an RC dendrite, allowing for so-called triadic inhibition. Taking advantage of a recently developed biophysically detailed multicompartmental model for an IN, we here investigate putative effects of these different inhibitory actions of INs, i.e., triadic inhibition and standard axonal inhibition, on the response properties of RCs. We compute and investigate so-called area-response curves, that is, trial-averaged visual spike responses vs. spot size, for circular flashing spots in a network of RCs and INs. The model parameters are grossly tuned to give results in qualitative accordance with previous in vivo data of responses to such stimuli for cat GCs and RCs. We particularly investigate how the model ingredients affect salient response properties such as the receptive-field center size of RCs and INs, maximal responses and center-surround antagonisms. For example, while triadic inhibition not involving firing of IN action potentials was found to provide only a non-linear gain control of the conversion of input spikes to output spikes by RCs, axonal inhibition was in contrast found to substantially affect the receptive-field center size: the larger the inhibition, the more the RC center size shrinks compared to the GC providing the feedforward excitation. Thus, a possible role of the different inhibitory actions from INs to RCs in the dLGN circuit is to provide separate mechanisms for overall gain control (direct triadic inhibition) and regulation of spatial resolution (axonal inhibition) of visual signals sent to cortex.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




Similar articles
-
Biophysical network modeling of the dLGN circuit: Effects of cortical feedback on spatial response properties of relay cells.PLoS Comput Biol. 2018 Jan 29;14(1):e1005930. doi: 10.1371/journal.pcbi.1005930. eCollection 2018 Jan. PLoS Comput Biol. 2018. PMID: 29377888 Free PMC article.
-
Spatial summation and center-surround antagonism in the receptive field of single units in the dorsal lateral geniculate nucleus of cat: comparison with retinal input.Vis Neurosci. 2000 Nov-Dec;17(6):855-70. doi: 10.1017/s0952523800176059. Vis Neurosci. 2000. PMID: 11193102
-
Mathematical models for the spatial receptive-field organization of nonlagged X-cells in dorsal lateral geniculate nucleus of cat.Vis Neurosci. 2000 Nov-Dec;17(6):871-85. doi: 10.1017/s0952523800176060. Vis Neurosci. 2000. PMID: 11193103
-
How much feedback from visual cortex to lateral geniculate nucleus in cat: a perspective.Vis Neurosci. 2004 Jul-Aug;21(4):487-500. doi: 10.1017/S0952523804214018. Vis Neurosci. 2004. PMID: 15579216 Review.
-
Organization of the dorsal lateral geniculate nucleus in the mouse.Vis Neurosci. 2017 Jan;34:E008. doi: 10.1017/S0952523817000062. Vis Neurosci. 2017. PMID: 28965501 Free PMC article. Review.
Cited by
-
Firing-rate based network modeling of the dLGN circuit: Effects of cortical feedback on spatiotemporal response properties of relay cells.PLoS Comput Biol. 2018 May 17;14(5):e1006156. doi: 10.1371/journal.pcbi.1006156. eCollection 2018 May. PLoS Comput Biol. 2018. PMID: 29771919 Free PMC article.
-
Morphology, physiology and synaptic connectivity of local interneurons in the mouse somatosensory thalamus.J Physiol. 2021 Nov;599(22):5085-5101. doi: 10.1113/JP281711. Epub 2021 Oct 23. J Physiol. 2021. PMID: 34591324 Free PMC article.
-
Multimodal Modeling of Neural Network Activity: Computing LFP, ECoG, EEG, and MEG Signals With LFPy 2.0.Front Neuroinform. 2018 Dec 18;12:92. doi: 10.3389/fninf.2018.00092. eCollection 2018. Front Neuroinform. 2018. PMID: 30618697 Free PMC article.
-
Biophysical network modeling of the dLGN circuit: Effects of cortical feedback on spatial response properties of relay cells.PLoS Comput Biol. 2018 Jan 29;14(1):e1005930. doi: 10.1371/journal.pcbi.1005930. eCollection 2018 Jan. PLoS Comput Biol. 2018. PMID: 29377888 Free PMC article.
-
State-dependent modulation of thalamocortical oscillations by gamma light flicker with different frequencies, intensities, and duty cycles.Front Neuroinform. 2022 Aug 23;16:968907. doi: 10.3389/fninf.2022.968907. eCollection 2022. Front Neuroinform. 2022. PMID: 36081653 Free PMC article.
References
-
- Sherman S, Guillery R. Exploring the Thalamus. New York: Academic; 2001.
-
- Mastronarde DN. Two classes of single-input X-cells in cat lateral geniculate nucleus. I. Receptive-field properties and classification of cells. Journal of Neurophysiology. 1987;57:357–380. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

