Lipoprotein(a) and oxidized phospholipids in calcific aortic valve stenosis
- PMID: 27205885
- PMCID: PMC4956483
- DOI: 10.1097/HCO.0000000000000300
Lipoprotein(a) and oxidized phospholipids in calcific aortic valve stenosis
Abstract
Purpose of review: As the incidence of calcific aortic valve stenosis increases with the aging of the population, improved understanding and novel therapies to reduce its progression and need for aortic valve replacement are urgently needed.
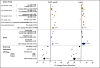
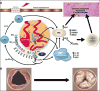
Recent findings: Lipoprotein(a) is the only monogenetic risk factor for calcific aortic stenosis. Elevated levels are a strong, causal, independent risk factor, as demonstrated in epidemiological, genome-wide association studies and Mendelian randomization studies. Lipoprotein(a) is the major lipoprotein carrier of oxidized phospholipids, which are proinflammatory and promote calcification of vascular cells, two key pathophysiological drivers of aortic stenosis. Elevated plasma lipoprotein(a) and oxidized phospholipids predict progression of pre-existing aortic stenosis and need for aortic valve replacement. The failure of statin trials in pre-existing aortic stenosis may be partially due to an increase in lipoprotein(a) and oxidized phospholipid levels caused by statins. Antisense oligonucleotides targeted to apo(a) are in Phase 2 clinical development and shown to lower both lipoprotein(a) and oxidized phospholipids.
Summary: Lipoprotein(a) and oxidized phospholipids are key therapeutic targets in calcific aortic stenosis. Strategies aimed at potent lipoprotein(a) lowering to normalize levels and/or to suppress the proinflammatory effects of oxidized phospholipids may prevent progression of this disease.
Conflict of interest statement
ST is a co-inventor of and receives royalties from patents or patent applications owned by the University of California San Diego and has a dual appointment at UCSD and Ionis Pharmaceuticals, Inc. The other authors report no conflicts.
Figures
References
-
- Otto CM, Prendergast B. Aortic-valve stenosis–from patients at risk to severe valve obstruction. N Engl J Med. 2014;371:744–756. - PubMed
-
- Pawade TA, Newby DE, Dweck MR. Calcification in Aortic Stenosis. The Skeleton Key. J Am Coll Cardiol. 2015;66:561–577. Excellent review article on the role of calcification in aortic stenosis. - PubMed
-
- Cowell SJ, Newby DE, Prescott RJ, et al. A randomized trial of intensive lipid-lowering therapy in calcific aortic stenosis. N Engl J Med. 2005;352:2389–2397. - PubMed
-
- Rossebo AB, Pedersen TR, Boman K, et al. Intensive lipid lowering with simvastatin and ezetimibe in aortic stenosis. N Engl J Med. 2008;359:1343–1356. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

