TOR complex 2 localises to the cytokinetic actomyosin ring and controls the fidelity of cytokinesis
- PMID: 27206859
- PMCID: PMC4958305
- DOI: 10.1242/jcs.190124
TOR complex 2 localises to the cytokinetic actomyosin ring and controls the fidelity of cytokinesis
Abstract
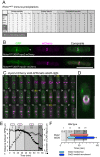
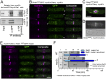
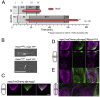
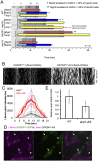
The timing of cell division is controlled by the coupled regulation of growth and division. The target of rapamycin (TOR) signalling network synchronises these processes with the environmental setting. Here, we describe a novel interaction of the fission yeast TOR complex 2 (TORC2) with the cytokinetic actomyosin ring (CAR), and a novel role for TORC2 in regulating the timing and fidelity of cytokinesis. Disruption of TORC2 or its localisation results in defects in CAR morphology and constriction. We provide evidence that the myosin II protein Myp2 and the myosin V protein Myo51 play roles in recruiting TORC2 to the CAR. We show that Myp2 and TORC2 are co-dependent upon each other for their normal localisation to the cytokinetic machinery. We go on to show that TORC2-dependent phosphorylation of actin-capping protein 1 (Acp1, a known regulator of cytokinesis) controls CAR stability, modulates Acp1-Acp2 (the equivalent of the mammalian CAPZA-CAPZB) heterodimer formation and is essential for survival upon stress. Thus, TORC2 localisation to the CAR, and TORC2-dependent Acp1 phosphorylation contributes to timely control and the fidelity of cytokinesis and cell division.
Keywords: Acp1; CAPZA; Myosin II; Myosin V; Rictor; Schizosaccharomyces pombe; TOR; TORC2.
© 2016. Published by The Company of Biologists Ltd.
Conflict of interest statement
The authors declare no competing or financial interests.
Figures


Similar articles
-
Three myosins contribute uniquely to the assembly and constriction of the fission yeast cytokinetic contractile ring.Curr Biol. 2015 Aug 3;25(15):1955-65. doi: 10.1016/j.cub.2015.06.018. Epub 2015 Jul 2. Curr Biol. 2015. PMID: 26144970 Free PMC article.
-
Role of the two type II myosins, Myo2 and Myp2, in cytokinetic actomyosin ring formation and function in fission yeast.Cell Motil Cytoskeleton. 2003 Mar;54(3):208-16. doi: 10.1002/cm.10093. Cell Motil Cytoskeleton. 2003. PMID: 12589679
-
Cytokinesis in fission yeast: a myosin pas de deux.Microsc Res Tech. 2000 Apr 15;49(2):152-60. doi: 10.1002/(SICI)1097-0029(20000415)49:2<152::AID-JEMT7>3.0.CO;2-7. Microsc Res Tech. 2000. PMID: 10816254 Review.
-
Actin turnover ensures uniform tension distribution during cytokinetic actomyosin ring contraction.Mol Biol Cell. 2019 Apr 1;30(8):933-941. doi: 10.1091/mbc.E18-08-0511. Epub 2019 Feb 13. Mol Biol Cell. 2019. PMID: 30759055 Free PMC article.
-
Regulation of contractile ring formation and septation in Schizosaccharomyces pombe.Curr Opin Microbiol. 2015 Dec;28:46-52. doi: 10.1016/j.mib.2015.08.001. Epub 2015 Sep 3. Curr Opin Microbiol. 2015. PMID: 26340438 Free PMC article. Review.
Cited by
-
TOR Complex 2- independent mutations in the regulatory PIF pocket of Gad8AKT1/SGK1 define separate branches of the stress response mechanisms in fission yeast.PLoS Genet. 2020 Nov 2;16(11):e1009196. doi: 10.1371/journal.pgen.1009196. eCollection 2020 Nov. PLoS Genet. 2020. PMID: 33137119 Free PMC article.
-
Ste12/Fab1 phosphatidylinositol-3-phosphate 5-kinase is required for nitrogen-regulated mitotic commitment and cell size control.PLoS One. 2017 Mar 8;12(3):e0172740. doi: 10.1371/journal.pone.0172740. eCollection 2017. PLoS One. 2017. PMID: 28273166 Free PMC article.
-
The TOR Signaling Pathway in Spatial and Temporal Control of Cell Size and Growth.Front Cell Dev Biol. 2017 Jun 7;5:61. doi: 10.3389/fcell.2017.00061. eCollection 2017. Front Cell Dev Biol. 2017. PMID: 28638821 Free PMC article. Review.
-
A single-headed fission yeast myosin V transports actin in a tropomyosin-dependent manner.J Cell Biol. 2016 Jul 18;214(2):167-79. doi: 10.1083/jcb.201511102. Epub 2016 Jul 11. J Cell Biol. 2016. PMID: 27432898 Free PMC article.
-
Nickel induced cell impairments are negatively regulated by the Tor1 kinase in Schizosaccharomyces pombe.World J Microbiol Biotechnol. 2021 Aug 30;37(10):165. doi: 10.1007/s11274-021-03130-2. World J Microbiol Biotechnol. 2021. PMID: 34458935
References
-
- Atkin J., Halova L., Ferguson J., Hitchin J. R., Lichawska-Cieslar A., Jordan A. M., Pines J., Wellbrock C. and Petersen J. (2014). Torin1-mediated TOR kinase inhibition reduces Wee1 levels and advances mitotic commitment in fission yeast and HeLa cells. J. Cell Sci. 127, 1346-1356. 10.1242/jcs.146373 - DOI - PMC - PubMed
-
- Bähler J., Wu J.-Q., Longtine M. S., Shah N. G., McKenzie A. III, Steever A. B., Wach A., Philippsen P. and Pringle J. R. (1998). Heterologous modules for efficient and versatile PCR-based gene targeting in Schizosaccharomyces pombe. Yeast 14, 943-951. 10.1002/(SICI)1097-0061(199807)14:10<943::AID-YEA292>3.0.CO;2-Y - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

