Setting Occupational Exposure Limits for Genotoxic Substances in the Pharmaceutical Industry
- PMID: 27207978
- PMCID: PMC4914798
- DOI: 10.1093/toxsci/kfw028
Setting Occupational Exposure Limits for Genotoxic Substances in the Pharmaceutical Industry
Abstract
In the pharmaceutical industry, genotoxic drug substances are developed for life-threatening indications such as cancer. Healthy employees handle these substances during research, development, and manufacturing; therefore, safe handling of genotoxic substances is essential. When an adequate preclinical dataset is available, a risk-based decision related to exposure controls for manufacturing is made following a determination of safe health-based limits, such as an occupational exposure limit (OEL). OELs are calculated for substances based on a threshold dose-response once a threshold is identified. In this review, we present examples of genotoxic mechanisms where thresholds can be demonstrated and OELs can be calculated, including a holistic toxicity assessment. We also propose a novel approach for inhalation Threshold of Toxicological Concern (TTC) limit for genotoxic substances in cases where the database is not adequate to determine a threshold.
Keywords: TTC; acceptable risk; genotoxicity; occupational exposure limit; threshold.
© The Author 2016. Published by Oxford University Press on behalf of the Society of Toxicology. All rights reserved. For Permissions, please e-mail: journals.permissions@oup.com.
Figures
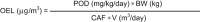
References
-
- Aydemir N., Celikler S., Bilaloğlu R. (2005). In vitro genotoxic effects of the anticancer drug gemcitabine in human lymphocytes. Mutat. Res. 582, 35–41. - PubMed
-
- Bercu J., Morinello E., Sehner C., Shipp B., Weideman P. (2016). Point of departure (PoD) selection for the derivation of acceptable daily exposure (ADE) value for Active Pharmaceutical Ingredients (APIs). Regul. Toxicol. Pharmacol. - PubMed
-
- Bundesanstalt für Arbeitsschutz und Arbeitsmedizin (BAuA) (2014). Techniche Regeln fur Gefarstoffe, TRGS 910: Risikobezogenes Maßnahmenkonzept für Tätigkeiten mit krebserzeugenden Gefahrstoffen. GMBI 64, 1313
-
- Cammerer Z., Schumacher M. M., Kirsch-Volders M., Suter W., Elhajouji A. (2010). Flow cytometry peripheral blood micronucleus test in vivo: Determination of potential thresholds for aneuploidy induced by spindle poisons. Environ. Mol. Mutagen. 51, 278–284. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

