Starch Biosynthesis in Guard Cells But Not in Mesophyll Cells Is Involved in CO2-Induced Stomatal Closing
- PMID: 27208296
- PMCID: PMC4902578
- DOI: 10.1104/pp.15.01662
Starch Biosynthesis in Guard Cells But Not in Mesophyll Cells Is Involved in CO2-Induced Stomatal Closing
Abstract
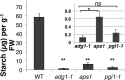
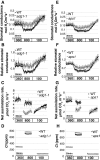
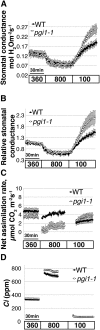
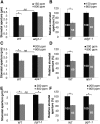
Starch metabolism is involved in stomatal movement regulation. However, it remains unknown whether starch-deficient mutants affect CO2-induced stomatal closing and whether starch biosynthesis in guard cells and/or mesophyll cells is rate limiting for high CO2-induced stomatal closing. Stomatal responses to [CO2] shifts and CO2 assimilation rates were compared in Arabidopsis (Arabidopsis thaliana) mutants that were either starch deficient in all plant tissues (ADP-Glc-pyrophosphorylase [ADGase]) or retain starch accumulation in guard cells but are starch deficient in mesophyll cells (plastidial phosphoglucose isomerase [pPGI]). ADGase mutants exhibited impaired CO2-induced stomatal closure, but pPGI mutants did not, showing that starch biosynthesis in guard cells but not mesophyll functions in CO2-induced stomatal closing. Nevertheless, starch-deficient ADGase mutant alleles exhibited partial CO2 responses, pointing toward a starch biosynthesis-independent component of the response that is likely mediated by anion channels. Furthermore, whole-leaf CO2 assimilation rates of both ADGase and pPGI mutants were lower upon shifts to high [CO2], but only ADGase mutants caused impairments in CO2-induced stomatal closing. These genetic analyses determine the roles of starch biosynthesis for high CO2-induced stomatal closing.
© 2016 American Society of Plant Biologists. All Rights Reserved.
Figures


Similar articles
-
Starch biosynthesis by AGPase, but not starch degradation by BAM1/3 and SEX1, is rate-limiting for CO2 -regulated stomatal movements under short-day conditions.FEBS Lett. 2018 Aug;592(16):2739-2759. doi: 10.1002/1873-3468.13198. Epub 2018 Aug 10. FEBS Lett. 2018. PMID: 30025149
-
Plastidic phosphoglucose isomerase is an important determinant of starch accumulation in mesophyll cells, growth, photosynthetic capacity, and biosynthesis of plastidic cytokinins in Arabidopsis.PLoS One. 2015 Mar 26;10(3):e0119641. doi: 10.1371/journal.pone.0119641. eCollection 2015. PLoS One. 2015. PMID: 25811607 Free PMC article.
-
Guard cell photosynthesis is critical for stomatal turgor production, yet does not directly mediate CO2 - and ABA-induced stomatal closing.Plant J. 2015 Aug;83(4):567-81. doi: 10.1111/tpj.12916. Epub 2015 Jul 22. Plant J. 2015. PMID: 26096271 Free PMC article.
-
New approaches to the biology of stomatal guard cells.Plant Cell Physiol. 2014 Feb;55(2):241-50. doi: 10.1093/pcp/pct145. Epub 2013 Oct 7. Plant Cell Physiol. 2014. PMID: 24104052 Free PMC article. Review.
-
Abscisic acid and CO2 signalling via calcium sensitivity priming in guard cells, new CDPK mutant phenotypes and a method for improved resolution of stomatal stimulus-response analyses.Ann Bot. 2012 Jan;109(1):5-17. doi: 10.1093/aob/mcr252. Epub 2011 Oct 12. Ann Bot. 2012. PMID: 21994053 Free PMC article. Review.
Cited by
-
Arabidopsis Sucrose Synthase 3 (SUS3) regulates starch accumulation in guard cells at the end of day.Plant Signal Behav. 2023 Dec 31;18(1):2171614. doi: 10.1080/15592324.2023.2171614. Plant Signal Behav. 2023. PMID: 36774587 Free PMC article.
-
The First Line of Defense: Receptor-like Protein Kinase-Mediated Stomatal Immunity.Int J Mol Sci. 2021 Dec 29;23(1):343. doi: 10.3390/ijms23010343. Int J Mol Sci. 2021. PMID: 35008769 Free PMC article. Review.
-
Arabidopsis guard cell chloroplasts import cytosolic ATP for starch turnover and stomatal opening.Nat Commun. 2022 Feb 3;13(1):652. doi: 10.1038/s41467-022-28263-2. Nat Commun. 2022. PMID: 35115512 Free PMC article.
-
Starch biosynthesis in guard cells has features of both autotrophic and heterotrophic tissues.Plant Physiol. 2022 Jun 1;189(2):541-556. doi: 10.1093/plphys/kiac087. Plant Physiol. 2022. PMID: 35238373 Free PMC article.
-
Responses of olive plants exposed to different irrigation treatments in combination with heat shock: physiological and molecular mechanisms during exposure and recovery.Planta. 2019 May;249(5):1583-1598. doi: 10.1007/s00425-019-03109-2. Epub 2019 Feb 15. Planta. 2019. PMID: 30771046
References
-
- Allaway WG, Setterfi G (1972) Ultrastructural observations on guard cells of Vicia faba and Allium porrum. Can J Bot 50: 1405
-
- Antunes WC, Provart NJ, Williams TCR, Loureiro ME (2012) Changes in stomatal function and water use efficiency in potato plants with altered sucrolytic activity. Plant Cell Environ 35: 747–759 - PubMed
-
- Bahaji A, Li J, Ovecka M, Ezquer I, Muñoz FJ, Baroja-Fernández E, Romero JM, Almagro G, Montero M, Hidalgo M, Sesma MT, Pozueta-Romero J (2011) Arabidopsis thaliana mutants lacking ADP-glucose pyrophosphorylase accumulate starch and wild-type ADP-glucose content: further evidence for the occurrence of important sources, other than ADP-glucose pyrophosphorylase, of ADP-glucose linked to leaf starch biosynthesis. Plant Cell Physiol 52: 1162–1176 - PubMed
-
- Bahaji A, Sánchez-López AM, De Diego N, Muñoz FJ, Baroja-Fernández E, Li J, Ricarte-Bermejo A, Baslam M, Aranjuelo I, Almagro G, et al. (2015) Plastidic phosphoglucose isomerase is an important determinant of starch accumulation in mesophyll cells, growth, photosynthetic capacity, and biosynthesis of plastidic cytokinins in Arabidopsis. PLoS One 10: e0119641. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

