A wearable chemical-electrophysiological hybrid biosensing system for real-time health and fitness monitoring
- PMID: 27212140
- PMCID: PMC4879240
- DOI: 10.1038/ncomms11650
A wearable chemical-electrophysiological hybrid biosensing system for real-time health and fitness monitoring
Abstract
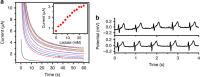
Flexible, wearable sensing devices can yield important information about the underlying physiology of a human subject for applications in real-time health and fitness monitoring. Despite significant progress in the fabrication of flexible biosensors that naturally comply with the epidermis, most designs measure only a small number of physical or electrophysiological parameters, and neglect the rich chemical information available from biomarkers. Here, we introduce a skin-worn wearable hybrid sensing system that offers simultaneous real-time monitoring of a biochemical (lactate) and an electrophysiological signal (electrocardiogram), for more comprehensive fitness monitoring than from physical or electrophysiological sensors alone. The two sensing modalities, comprising a three-electrode amperometric lactate biosensor and a bipolar electrocardiogram sensor, are co-fabricated on a flexible substrate and mounted on the skin. Human experiments reveal that physiochemistry and electrophysiology can be measured simultaneously with negligible cross-talk, enabling a new class of hybrid sensing devices.
Figures



References
-
- Kim D.-H., Ghaffari R., Lu N. & Rogers J. A. Flexible and stretchable electronics for biointegrated devices. Annu. Rev. Biomed. Eng. 14, 113–128 (2012). - PubMed
-
- Bandodkar A. J. & Wang J. Non-invasive wearable electrochemical sensors: a review. Trends Biotechnol. 32, 363–371 (2014). - PubMed
-
- Chuang M. C. et al. Flexible thick-film glucose biosensor: influence of mechanical bending on the performance. Talanta 81, 15–19 (2010). - PubMed
-
- Kim D.-H. et al. Epidermal electronics. Science 333, 838–844 (2011). - PubMed
-
- Merritt C. R., Nagle H. T. & Grant E. Textile-based capacitive sensors for respiration monitoring. IEEE Sens. J. 9, 71–78 (2009).
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

