Corticosteroids Are Essential for Maintaining Cardiovascular Function in Male Mice
- PMID: 27219275
- PMCID: PMC4929548
- DOI: 10.1210/en.2015-1604
Corticosteroids Are Essential for Maintaining Cardiovascular Function in Male Mice
Abstract
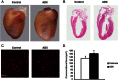
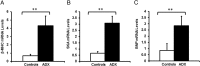
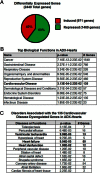
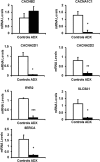
Activation of the hypothalamic-pituitary-adrenal axis results in the release of hormones from the adrenal glands, including glucocorticoids and mineralocorticoids. The physiological association between corticosteroids and cardiac disease is becoming increasingly recognized; however, the mechanisms underlying this association are not well understood. To determine the biological effects of corticosteroids on the heart, we investigated the impact of adrenalectomy in C57BL/6 male mice. Animals were adrenalectomized (ADX) at 1 month of age and maintained for 3-6 months after surgery to evaluate the effects of long-term adrenalectomy on cardiac function. Morphological evaluation suggested that ADX mice showed significantly enlarged hearts compared with age-matched intact controls. These changes in morphology correlated with deficits in left ventricular (LV) function and electrocardiogram (ECG) abnormalities in ADX mice. Correlating with these functional defects, gene expression analysis of ADX hearts revealed aberrant expression of a large cohort of genes associated with cardiac hypertrophy and arrhythmia. Combined corticosterone and aldosterone replacement treatment prevented the emergence of cardiac abnormalities in ADX mice, whereas corticosterone replacement prevented the effects of adrenalectomy on LV function but did not block the emergence of ECG alterations. Aldosterone replacement did not preserve the LV function but prevented ECG abnormalities. Together, the data indicate that adrenal glucocorticoids and mineralocorticoids either directly or indirectly have selective effects in the heart and their signaling pathways are essential in maintaining normal cardiac function.
Figures




Comment in
-
Endocrine Affairs of the Heart.Endocrinology. 2016 Jul;157(7):2578-82. doi: 10.1210/en.2016-1375. Endocrinology. 2016. PMID: 27363869 No abstract available.
Similar articles
-
Adrenalectomy amplifies aldosterone induced injury in cardiovascular tissue: an effect attenuated by adrenally derived steroids.Steroids. 2013 Mar;78(3):347-55. doi: 10.1016/j.steroids.2012.12.007. Epub 2012 Dec 31. Steroids. 2013. PMID: 23287650
-
Regulatory mechanism of hypothalamo-pituitary-adrenal (HPA) axis and neuronal changes after adrenalectomy in type 2 diabetes.J Chem Neuroanat. 2010 Oct;40(2):130-9. doi: 10.1016/j.jchemneu.2010.05.003. Epub 2010 May 21. J Chem Neuroanat. 2010. PMID: 20472052
-
Role of the dorsomedial hypothalamus in glucocorticoid-mediated feedback inhibition of the hypothalamic-pituitary-adrenal axis.Stress. 2015 Jan;18(1):76-87. doi: 10.3109/10253890.2015.1004537. Epub 2015 Feb 17. Stress. 2015. PMID: 25556980 Free PMC article.
-
Regulation of nephron acidification by corticosteroids.Braz J Med Biol Res. 1997 Apr;30(4):479-86. doi: 10.1590/s0100-879x1997000400008. Braz J Med Biol Res. 1997. PMID: 9251768 Review.
-
Extra-adrenal mineralocorticoids and cardiovascular tissue.J Mol Cell Cardiol. 1999 Jun;31(6):1175-84. doi: 10.1006/jmcc.1999.0963. J Mol Cell Cardiol. 1999. PMID: 10371693 Review.
Cited by
-
Plasma renin levels are associated with cardiac function in primary adrenal insufficiency.Endocrine. 2019 Aug;65(2):399-407. doi: 10.1007/s12020-019-01974-1. Epub 2019 Jun 8. Endocrine. 2019. PMID: 31177424 Free PMC article.
-
Revisiting treatment-related cardiotoxicity in patients with malignant lymphoma-a review and prospects for the future.Front Cardiovasc Med. 2023 Aug 30;10:1243531. doi: 10.3389/fcvm.2023.1243531. eCollection 2023. Front Cardiovasc Med. 2023. PMID: 37711551 Free PMC article. Review.
-
The Glucocorticoid Receptor NR3C1 in Testicular Peritubular Cells is Developmentally Regulated and Linked to the Smooth Muscle-Like Cellular Phenotype.J Clin Med. 2020 Mar 31;9(4):961. doi: 10.3390/jcm9040961. J Clin Med. 2020. PMID: 32244354 Free PMC article.
-
Hypothalamic-Pituitary-Adrenal Axis Modulation of Glucocorticoids in the Cardiovascular System.Int J Mol Sci. 2017 Oct 16;18(10):2150. doi: 10.3390/ijms18102150. Int J Mol Sci. 2017. PMID: 29035323 Free PMC article. Review.
-
Getting to the heart of intracellular glucocorticoid regeneration: 11β-HSD1 in the myocardium.J Mol Endocrinol. 2017 Jan;58(1):R1-R13. doi: 10.1530/JME-16-0128. Epub 2016 Aug 23. J Mol Endocrinol. 2017. PMID: 27553202 Free PMC article. Review.
References
-
- Funder JW, Pearce PT, Smith R, Smith AI. Mineralocorticoid action: target tissue specificity is enzyme, not receptor, mediated. Science. 1988;242:583–585. - PubMed
-
- Meinel S, Gekle M, Grossmann C. Mineralocorticoid receptor signaling: crosstalk with membrane receptors and other modulators. Steroids. 2014;91:3–10. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

