Magnetoencephalographic signatures of insular epileptic spikes based on functional connectivity
- PMID: 27220112
- PMCID: PMC6867448
- DOI: 10.1002/hbm.23238
Magnetoencephalographic signatures of insular epileptic spikes based on functional connectivity
Abstract
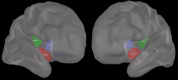
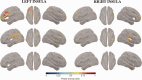
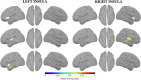
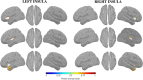
Failure to recognize insular cortex seizures has recently been identified as a cause of epilepsy surgeries targeting the temporal, parietal, or frontal lobe. Such failures are partly due to the fact that current noninvasive localization techniques fare poorly in recognizing insular epileptic foci. Our group recently demonstrated that magnetoencephalography (MEG) is sensitive to epileptiform spikes generated by the insula. In this study, we assessed the potential of distributed source imaging and functional connectivity analyses to distinguish insular networks underlying the generation of spikes. Nineteen patients with operculo-insular epilepsy were investigated. Each patient underwent MEG as well as T1-weighted magnetic resonance imaging (MRI) as part of their standard presurgical evaluation. Cortical sources of MEG spikes were reconstructed with the maximum entropy on the mean algorithm, and their time courses served to analyze source functional connectivity. The results indicate that the anterior and posterior subregions of the insula have specific patterns of functional connectivity mainly involving frontal and parietal regions, respectively. In addition, while their connectivity patterns are qualitatively similar during rest and during spikes, couplings within these networks are much stronger during spikes. These results show that MEG can establish functional connectivity-based signatures that could help in the diagnosis of different subtypes of insular cortex epilepsy. Hum Brain Mapp 37:3250-3261, 2016. © 2016 Wiley Periodicals, Inc.
Keywords: brain disorders; brain imaging; cortical phase synchronization; epilepsies; nonparametric statistics; partial.
© 2016 Wiley Periodicals, Inc.
Figures


Similar articles
-
The role of magnetoencephalography in the presurgical evaluation of patients with MRI-negative operculo-insular epilepsy.Seizure. 2018 Oct;61:104-110. doi: 10.1016/j.seizure.2018.07.005. Epub 2018 Aug 13. Seizure. 2018. PMID: 30125861
-
The utility of magnetoencephalography in the presurgical evaluation of refractory insular epilepsy.Epilepsia. 2013 Nov;54(11):1950-9. doi: 10.1111/epi.12376. Epub 2013 Sep 30. Epilepsia. 2013. PMID: 24117237
-
Detection and localization of interictal ripples with magnetoencephalography in the presurgical evaluation of drug-resistant insular epilepsy.Brain Res. 2019 Mar 1;1706:147-156. doi: 10.1016/j.brainres.2018.11.006. Epub 2018 Nov 5. Brain Res. 2019. PMID: 30408475
-
Multimodal investigation of epileptic networks: The case of insular cortex epilepsy.Prog Brain Res. 2016;226:1-33. doi: 10.1016/bs.pbr.2016.04.004. Epub 2016 May 6. Prog Brain Res. 2016. PMID: 27323937 Review.
-
Electroencephalography, magnetoencephalography and source localization: their value in epilepsy.Curr Opin Neurol. 2018 Apr;31(2):176-183. doi: 10.1097/WCO.0000000000000545. Curr Opin Neurol. 2018. PMID: 29432218 Review.
Cited by
-
Magnetoencephalography in Preoperative Epileptic Foci Localization: Enlightenment from Cognitive Studies.Front Comput Neurosci. 2017 Jun 28;11:58. doi: 10.3389/fncom.2017.00058. eCollection 2017. Front Comput Neurosci. 2017. PMID: 28701945 Free PMC article. Review.
-
A Case of Hypermotor Seizures in Posterior Insular Cortex Epilepsy.Cureus. 2023 Mar 17;15(3):e36280. doi: 10.7759/cureus.36280. eCollection 2023 Mar. Cureus. 2023. PMID: 37075168 Free PMC article.
-
Structural Connectivity Alterations in Operculo-Insular Epilepsy.Brain Sci. 2021 Aug 5;11(8):1041. doi: 10.3390/brainsci11081041. Brain Sci. 2021. PMID: 34439659 Free PMC article.
-
Cortical thickness analysis in operculo-insular epilepsy.Neuroimage Clin. 2018 May 31;19:727-733. doi: 10.1016/j.nicl.2018.05.033. eCollection 2018. Neuroimage Clin. 2018. PMID: 30003025 Free PMC article.
-
Practical Fundamentals of Clinical MEG Interpretation in Epilepsy.Front Neurol. 2021 Oct 14;12:722986. doi: 10.3389/fneur.2021.722986. eCollection 2021. Front Neurol. 2021. PMID: 34721261 Free PMC article. Review.
References
-
- Aghakhani Y, Rosati A, Dubeau F, Olivier A, Andermann F (2004): Patients with temporoparietal ictal symptoms and inferomesial EEG do not benefit from anterior temporal resection. Epilepsia 45:230–236. - PubMed
-
- Alarcon G, Garcia Seoane JJ, Binnie CD, Martin Miguel MC, Juler J, Polkey CE, Elwes RD, Ortiz Blasco JM (1997): Origin and propagation of interictal discharges in the acute electrocorticogram. Implications for pathophysiology and surgical treatment of temporal lobe epilepsy. Brain 120:2259–2282. - PubMed
-
- Amblard C, Lapalme E, Lina JM (2004): Biomagnetic source detection by maximum entropy and graphical models. IEEE Trans Biomed Eng 51:427–442. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

