Oxidative stress during erythropoietin hyporesponsiveness anemia at end stage renal disease: Molecular and biochemical studies
- PMID: 27222740
- PMCID: PMC4856826
- DOI: 10.1016/j.jare.2016.02.004
Oxidative stress during erythropoietin hyporesponsiveness anemia at end stage renal disease: Molecular and biochemical studies
Abstract
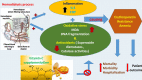
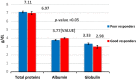
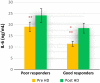
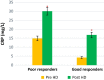
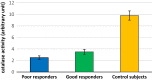
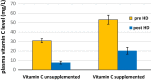
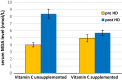
Inflammation and oxidative stress are two faces of one coin in end stage renal disease patients (ESRD) on maintenance hemodialysis. Their interconnection induces anemia complicated with erythropoietin hyporesponsiveness. The biochemical bases behind the resistance to erythropoietin therapy with frequent hemoglobinemia, oxidative stress and iron status have not been fully understood. Here two equal groups (40 patients each) of responders and non-responders to recombinant human erythropoietin therapy (higher than 300 IU/kg/wk of epoetin) were investigated. Hematological and biochemical analyses of collected blood and serum samples were performed along with serum electrophoretic protein footprinting. The leukocytic DNA fragmentation was used to evaluate the degree of oxidative insult. The good responders showed lower erythrocyte malondialdehyde (E-MDA) level and less DNA fragmentation of circulating leukocytes than poor responders with elevated hemoglobin, albumin, A/G ratio, total iron, and ferritin levels. Contrariwise, lower erythrocyte superoxide dismutase (E-SOD) and catalase activities in EPO poor responder group were noticed. Neither other serum constituents nor electrophoretic protein pattern showed any difference between the two groups. There were higher levels of inflammatory markers, interleukin-6 (IL6) and C-reactive protein (CRP) in EPO poor responder than good responder. The negative correlations between Hb and both IL6 and CRP levels in the present data remotely indicate a positive correlation between inflammatory markers and severity of anemia. A direct correlation between Hb and antioxidant enzymes (E-SOD and catalase) was noticed, while inverse correlation with E-MDA was recorded. The study proved that oral supplementation of vitamin C to ESRD patients might mitigate the previously elevated serum MDA level in these patients.
Keywords: Anemia of chronic disease; Erythropoietin resistance; Hemodialysis; Inflammatory markers; Oxidative stress; Vitamin C.
Figures



Similar articles
-
Effect of intravenous ascorbic acid in hemodialysis patients with EPO-hyporesponsive anemia and hyperferritinemia.Am J Kidney Dis. 2006 Apr;47(4):644-54. doi: 10.1053/j.ajkd.2005.12.025. Am J Kidney Dis. 2006. PMID: 16564942 Clinical Trial.
-
Nonalcoholic fatty liver disease (NAFLD)--is it a new marker of hyporesponsiveness to recombinant human erythropoietin in patients that are on chronic hemodialysis?Med Hypotheses. 2014 Dec;83(6):798-801. doi: 10.1016/j.mehy.2014.10.012. Epub 2014 Oct 22. Med Hypotheses. 2014. PMID: 25456789
-
Evaluation of Effect of Vitamin D Deficiency on Anemia and Erythropoietin Hyporesponsiveness in Patients of Chronic Kidney Disease.J Assoc Physicians India. 2017 Feb;65(2):38-42. J Assoc Physicians India. 2017. PMID: 28457030
-
[Erythropoietin-beta in the treatment of anemia in patients with chronic renal insufficiency].Med Pregl. 2001 May-Jun;54(5-6):235-40. Med Pregl. 2001. PMID: 11759218 Review. Croatian.
-
[Guidelines for the treatment of anemia in chronic renal failure].G Ital Nefrol. 2003 Sep-Oct;20 Suppl 24:S61-82. G Ital Nefrol. 2003. PMID: 14666504 Review. Italian.
Cited by
-
Biocompatibility of Polysulfone Hemodialysis Membranes and Its Mechanisms: Involvement of Fibrinogen and Its Integrin Receptors in Activation of Platelets and Neutrophils.Artif Organs. 2018 Sep;42(9):E246-E258. doi: 10.1111/aor.13268. Epub 2018 Sep 21. Artif Organs. 2018. PMID: 30239013 Free PMC article.
-
The Anti-Aging Effect of Erythropoietin via the ERK/Nrf2-ARE Pathway in Aging Rats.J Mol Neurosci. 2017 Mar;61(3):449-458. doi: 10.1007/s12031-017-0885-1. Epub 2017 Feb 6. J Mol Neurosci. 2017. PMID: 28168414
-
Oxidative Stress Parameters Can Predict the Response to Erythropoiesis-Stimulating Agents in Myelodysplastic Syndrome Patients.Front Cell Dev Biol. 2021 Jun 7;9:701328. doi: 10.3389/fcell.2021.701328. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34164406 Free PMC article.
-
Erythropoietin Resistance Development in Hemodialysis Patients: The Role of Oxidative Stress.Oxid Med Cell Longev. 2022 Apr 14;2022:9598211. doi: 10.1155/2022/9598211. eCollection 2022. Oxid Med Cell Longev. 2022. PMID: 35464768 Free PMC article.
-
Uremic leontiasis ossea due to secondary hyperparathyroidism complicated by vitamin C deficiency in a non-adherent chronic hemodialysis patient: A case report.Clin Nephrol Case Stud. 2019 Sep 9;7:54-59. doi: 10.5414/CNCS109788. eCollection 2019. Clin Nephrol Case Stud. 2019. PMID: 31508269 Free PMC article.
References
-
- Hörl W.H. Anaemia as a risk factor for chronic kidney disease. Arch Med Sci. 2009;5(3A):S421–S428.
-
- Astor B.C., Muntner P., Levin A., Eustace J.A., Coresh J. Association of kidney function with anemia: the Third National Health and Nutrition Examination Survey (1988–1994) Arch Intern Med. 2002;162:1401–1408. - PubMed
-
- McClellan W., Aronoff S.L., Bolton W.K., Hood S., Lorber D.L., Tang K.L. The prevalence of anemia in patients with chronic kidney disease. Curr Med Res Opin. 2004;20:1501–1510. - PubMed
-
- Dowling T.C. Prevalence, etiology, and consequences of anemia and clinical and economic benefits of anemia correction in patients with chronic kidney disease: an overview. Am J Health Syst Pharm. 2007;64(13 suppl 8):3–7. - PubMed
-
- Weiss G., Theurl I., Eder S., Koppelstaetter C., Kurz K., Sonnweber T. Serum hepcidin concentration in chronic haemodialysis patients: associations and effects of dialysis, iron and erythropoietin therapy. Eur J Clin Invest. 2009;39:883–890. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

