MCT4 as a potential therapeutic target for metastatic gastric cancer with peritoneal carcinomatosis
- PMID: 27224918
- PMCID: PMC5190039
- DOI: 10.18632/oncotarget.9523
MCT4 as a potential therapeutic target for metastatic gastric cancer with peritoneal carcinomatosis
Abstract
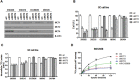
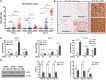
Monocarboxylate transporters (MCTs) play a major role in up-regulation of glycolysis and adaptation to acidosis. However, the role of MCTs in gastric cancer (GC) is not fully understood. We investigated the potential utilization of a new cancer therapy for GC. We characterized the expression patterns of the MCT isoforms 1, 2, and 4 and investigated the role of MCT in GC through in vitro and in vivo tests using siRNA targeting MCTs. In GC cell lines, MCT1, 2, and 4 were up-regulated with different expression levels; MCT1 and MCT4 were more widely expressed in GC cell lines compared with MCT2. Inhibition of MCTs by siRNA or AR-C155858 reduced cell viability and lactate uptake in GC cell lines. The effect of inhibition of MCTs on tumor growth was also confirmed in xenograft models. Furthermore, MCT inhibition in GC cells increased the sensitivity of cells to radiotherapy or chemotherapy. Compared with normal gastric tissue, no significant alterations of expression levels in tumors were identified for MCT1 and MCT2, whereas a significant increase in MCT4 expression was observed. Most importantly, MCT4 was highly overexpressed in malignant cells of acsites and its silencing resulted in reduced tumor cell proliferation and lactate uptake in malignant ascites. Our study suggests that MCT4 is a clinically relevant target in GC with peritoneal carcinomatosis.
Keywords: gastric cancer; glycolysis; monocarboxylate transporter; prognosis.
Conflict of interest statement
The authors have no potential conflicts of interest to declare.
Figures



Similar articles
-
MCT4 Expression Is a Potential Therapeutic Target in Colorectal Cancer with Peritoneal Carcinomatosis.Mol Cancer Ther. 2018 Apr;17(4):838-848. doi: 10.1158/1535-7163.MCT-17-0535. Epub 2018 Feb 26. Mol Cancer Ther. 2018. PMID: 29483215
-
CD147 subunit of lactate/H+ symporters MCT1 and hypoxia-inducible MCT4 is critical for energetics and growth of glycolytic tumors.Proc Natl Acad Sci U S A. 2011 Oct 4;108(40):16663-8. doi: 10.1073/pnas.1106123108. Epub 2011 Sep 19. Proc Natl Acad Sci U S A. 2011. PMID: 21930917 Free PMC article.
-
In Vitro and In Vivo Efficacy of the Monocarboxylate Transporter 1 Inhibitor AR-C155858 in the Murine 4T1 Breast Cancer Tumor Model.AAPS J. 2018 Nov 5;21(1):3. doi: 10.1208/s12248-018-0261-2. AAPS J. 2018. PMID: 30397860 Free PMC article.
-
Monocarboxylate transporter 1 and 4 inhibitors as potential therapeutics for treating solid tumours: A review with structure-activity relationship insights.Eur J Med Chem. 2020 Aug 1;199:112393. doi: 10.1016/j.ejmech.2020.112393. Epub 2020 May 1. Eur J Med Chem. 2020. PMID: 32388280 Review.
-
The expression of lactate transporters (MCT1 and MCT4) in heart and muscle.Eur J Appl Physiol. 2001 Nov;86(1):6-11. doi: 10.1007/s004210100516. Eur J Appl Physiol. 2001. PMID: 11820324 Review.
Cited by
-
Metabolic coupling and the Reverse Warburg Effect in cancer: Implications for novel biomarker and anticancer agent development.Semin Oncol. 2017 Jun;44(3):198-203. doi: 10.1053/j.seminoncol.2017.10.004. Epub 2017 Oct 10. Semin Oncol. 2017. PMID: 29248131 Free PMC article. Review.
-
Overexpression of monocarboxylate transporter 4 promotes the migration and invasion of non-carcinogenic L929 fibroblast cells.Oncol Lett. 2021 Jan;21(1):44. doi: 10.3892/ol.2020.12305. Epub 2020 Nov 17. Oncol Lett. 2021. PMID: 33262836 Free PMC article.
-
Correlations between glycolysis with clinical traits and immune function in bladder urothelial carcinoma.Biosci Rep. 2021 Feb 26;41(2):BSR20203982. doi: 10.1042/BSR20203982. Biosci Rep. 2021. PMID: 33558879 Free PMC article.
-
Lactylation-related gene signature accurately predicts prognosis and immunotherapy response in gastric cancer.Front Oncol. 2024 Nov 28;14:1485580. doi: 10.3389/fonc.2024.1485580. eCollection 2024. Front Oncol. 2024. PMID: 39669362 Free PMC article.
-
Lactate and lactylation in cancer.Signal Transduct Target Ther. 2025 Feb 12;10(1):38. doi: 10.1038/s41392-024-02082-x. Signal Transduct Target Ther. 2025. PMID: 39934144 Free PMC article. Review.
References
-
- Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90. - PubMed
-
- Kamangar F, Dores GM, Anderson WF. Patterns of cancer incidence, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J Clin Oncol. 2006;24:2137–2150. - PubMed
-
- Cunningham SC, Kamangar F, Kim MP, Hammoud S, Haque R, Maitra A, Montgomery E, Heitmiller RE, Choti MA, Lillemoe KD, Cameron JL, Yeo CJ, Schulick RD. Survival after gastric adenocarcinoma resection: eighteen-year experience at a single institution. J Gastrointest Surg. 2005;9:718–725. - PubMed
-
- Cristescu R, Lee J, Nebozhyn M, Kim KM, Ting JC, Wong SS, Liu J, Yue YG, Wang J, Yu K, Ye XS, Do IG, Liu S, et al. Molecular analysis of gastric cancer identifies subtypes associated with distinct clinical outcomes. Nat Med. 2015;21:449–456. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

