Adenomatous polyposis coli regulates radial axonal sorting and myelination in the PNS
- PMID: 27226321
- PMCID: PMC4958326
- DOI: 10.1242/dev.135913
Adenomatous polyposis coli regulates radial axonal sorting and myelination in the PNS
Abstract
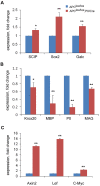
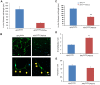
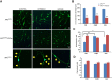
The tumor suppressor protein adenomatous polyposis coli (APC) is multifunctional - it participates in the canonical Wnt/β-catenin signal transduction pathway as well as modulating cytoskeleton function. Although APC is expressed by Schwann cells, the role that it plays in these cells and in the myelination of the peripheral nervous system (PNS) is unknown. Therefore, we used the Cre-lox approach to generate a mouse model in which APC expression is specifically eliminated from Schwann cells. These mice display hindlimb weakness and impaired axonal conduction in sciatic nerves. Detailed morphological analyses revealed that APC loss delays radial axonal sorting and PNS myelination. Furthermore, APC loss delays Schwann cell differentiation in vivo, which correlates with persistent activation of the Wnt signaling pathway and results in perturbed extension of Schwann cell processes and disrupted lamellipodia formation. In addition, APC-deficient Schwann cells display a transient diminution of proliferative capacity. Our data indicate that APC is required by Schwann cells for their timely differentiation to mature, myelinating cells and plays a crucial role in radial axonal sorting and PNS myelination.
Keywords: Adenomatous polyposis coli; PNS; Radial axonal sorting; Schwann cells; Wnt signaling.
© 2016. Published by The Company of Biologists Ltd.
Conflict of interest statement
The authors declare no competing or financial interests.
Figures
Similar articles
-
The Polarity Protein Pals1 Regulates Radial Sorting of Axons.J Neurosci. 2015 Jul 22;35(29):10474-84. doi: 10.1523/JNEUROSCI.1593-15.2015. J Neurosci. 2015. PMID: 26203142 Free PMC article.
-
Akt Regulates Axon Wrapping and Myelin Sheath Thickness in the PNS.J Neurosci. 2016 Apr 20;36(16):4506-21. doi: 10.1523/JNEUROSCI.3521-15.2016. J Neurosci. 2016. PMID: 27098694 Free PMC article.
-
Expression of laminin receptors in schwann cell differentiation: evidence for distinct roles.J Neurosci. 2003 Jul 2;23(13):5520-30. doi: 10.1523/JNEUROSCI.23-13-05520.2003. J Neurosci. 2003. PMID: 12843252 Free PMC article.
-
How Schwann Cells Sort Axons: New Concepts.Neuroscientist. 2016 Jun;22(3):252-65. doi: 10.1177/1073858415572361. Epub 2015 Feb 16. Neuroscientist. 2016. PMID: 25686621 Free PMC article. Review.
-
New insights into signaling during myelination in zebrafish.Curr Top Dev Biol. 2011;97:1-19. doi: 10.1016/B978-0-12-385975-4.00007-3. Curr Top Dev Biol. 2011. PMID: 22074600 Free PMC article. Review.
Cited by
-
αV integrins in Schwann cells promote attachment to axons, but are dispensable in vivo.Glia. 2021 Jan;69(1):91-108. doi: 10.1002/glia.23886. Epub 2020 Aug 3. Glia. 2021. PMID: 32744761 Free PMC article.
-
Schwann cell interactions during the development of the peripheral nervous system.Dev Neurobiol. 2021 Jul;81(5):464-489. doi: 10.1002/dneu.22744. Epub 2020 May 5. Dev Neurobiol. 2021. PMID: 32281247 Free PMC article. Review.
-
The bone transcription factor Osterix controls extracellular matrix- and node of Ranvier-related gene expression in oligodendrocytes.Neuron. 2024 Jan 17;112(2):247-263.e6. doi: 10.1016/j.neuron.2023.10.008. Epub 2023 Nov 3. Neuron. 2024. PMID: 37924811 Free PMC article.
-
Adenomatous Polyposis Coli (APC) in cell migration.Eur J Cell Biol. 2022 Jun-Aug;101(3):151228. doi: 10.1016/j.ejcb.2022.151228. Epub 2022 Apr 22. Eur J Cell Biol. 2022. PMID: 35483122 Free PMC article. Review.
-
Sensory neurons display cell-type-specific vulnerability to loss of neuron-glia interactions.Cell Rep. 2022 Jul 19;40(3):111130. doi: 10.1016/j.celrep.2022.111130. Cell Rep. 2022. PMID: 35858549 Free PMC article.
References
-
- Brosius Lutz A. (2014). Purification of Schwann cells from the neonatal and injured adult mouse peripheral nerve. In Purifying and Culturing Neural Cells: A Laboratory Manual (ed. Ben B. S. and Barres A.), pp. 177-188. Cold Spring Harbor Laboratory Press (in print). - PubMed
-
- Eccleston P. A., Mirsky R., Jessen K. R., Sommer I. and Schachner M. (1987). Postnatal development of rat peripheral nerves: an immunohistochemical study of membrane lipids common to non-myelin forming Schwann cells, myelin forming Schwann cells and oligodendrocytes. Brain Res. Dev Brain Res. 432, 249-256. 10.1016/0165-3806(87)90049-6 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

