Synergistic inhibition of colon cancer cell growth with nanoemulsion-loaded paclitaxel and PI3K/mTOR dual inhibitor BEZ235 through apoptosis
- PMID: 27226714
- PMCID: PMC4863683
- DOI: 10.2147/IJN.S100744
Synergistic inhibition of colon cancer cell growth with nanoemulsion-loaded paclitaxel and PI3K/mTOR dual inhibitor BEZ235 through apoptosis
Abstract
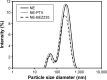
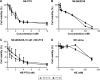
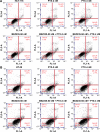
Colon cancer is the third most common cancer in the world, with drug resistance and metastasis being the major challenges to effective treatments. To overcome this, combination therapy with different chemotherapeutics is a common practice. In this study, we demonstrated that paclitaxel (PTX) together with BEZ235 exhibited a synergetic inhibition effect on colon cancer cell growth. Furthermore, nanoemulsion (NE)-loaded PTX and BEZ235 were more effective than the free drug, and a combination treatment of both NE drugs increased the efficiency of the treatments. BEZ235 pretreatment before adding PTX sensitized the cancer cells further, suggesting a synergistic inhibition effect through the phosphatidylinositol-3-kinases/protein kinase B/mammalian target of rapamycin pathway. The 50% inhibitory concentrations for BEZ235 were 127.1 nM and 145.0 nM and for PTX 9.7 nM and 9.5 nM for HCT-116 and HT-29 cells, respectively. When loaded with NE the 50% inhibitory concentrations for BEZ235 decreased to 52.6 nM and 55.6 nM and for PTX to 1.9 nM and 2.3 nM for HCT-116 and HT-29 cells, respectively. Combination treatment with 10 nM NE-BEZ235 and 0.6 nM and 1.78 nM NE-PTX could kill 50% of HCT-116 and HT-29, respectively. The cell death caused by the treatment was through apoptotic cell death, which coincided with decreased expression of anti-apoptotic protein B-cell lymphoma 2. Our data indicate that the combination therapy of PTX with the phosphatidylinositol-3-kinases/protein kinase B/mammalian target of rapamycin dual inhibitor BEZ235 using NE delivery may hold promise for a more effective approach for colon cancer treatment.
Keywords: chemotherapy; combination therapy; nanomedicine; signal pathway inhibitor.
Figures




Similar articles
-
Synergistic Inhibition of Drug-Resistant Colon Cancer Growth with PI3K/mTOR Dual Inhibitor BEZ235 and Nano-Emulsioned Paclitaxel via Reducing Multidrug Resistance and Promoting Apoptosis.Int J Nanomedicine. 2021 Mar 15;16:2173-2186. doi: 10.2147/IJN.S290731. eCollection 2021. Int J Nanomedicine. 2021. PMID: 33758505 Free PMC article.
-
Selective PI3K inhibition by BKM120 and BEZ235 alone or in combination with chemotherapy in wild-type and mutated human gastrointestinal cancer cell lines.Cancer Chemother Pharmacol. 2012 Jun;69(6):1601-15. doi: 10.1007/s00280-012-1869-z. Epub 2012 Apr 29. Cancer Chemother Pharmacol. 2012. PMID: 22543857
-
Dual PI3K/mTOR inhibitor BEZ235 as a promising therapeutic strategy against paclitaxel-resistant gastric cancer via targeting PI3K/Akt/mTOR pathway.Cell Death Dis. 2018 Jan 26;9(2):123. doi: 10.1038/s41419-017-0132-2. Cell Death Dis. 2018. PMID: 29374144 Free PMC article.
-
Co-treatment with BEZ235 enhances chemosensitivity of A549/DDP cells to cisplatin via inhibition of PI3K/Akt/mTOR signaling and downregulation of ERCC1 expression.Oncol Rep. 2018 Oct;40(4):2353-2362. doi: 10.3892/or.2018.6583. Epub 2018 Jul 20. Oncol Rep. 2018. PMID: 30066933
-
Dual blockade of PI3K/AKT/mTOR (NVP-BEZ235) and Ras/Raf/MEK (AZD6244) pathways synergistically inhibit growth of primary endometrioid endometrial carcinoma cultures, whereas NVP-BEZ235 reduces tumor growth in the corresponding xenograft models.Gynecol Oncol. 2015 Jul;138(1):165-73. doi: 10.1016/j.ygyno.2015.04.028. Epub 2015 Apr 28. Gynecol Oncol. 2015. PMID: 25933683
Cited by
-
PD-L1 Distribution and Perspective for Cancer Immunotherapy-Blockade, Knockdown, or Inhibition.Front Immunol. 2019 Aug 27;10:2022. doi: 10.3389/fimmu.2019.02022. eCollection 2019. Front Immunol. 2019. PMID: 31507611 Free PMC article. Review.
-
Synergistic antitumor effect of 5-fluorouracil and withaferin-A induces endoplasmic reticulum stress-mediated autophagy and apoptosis in colorectal cancer cells.Am J Cancer Res. 2020 Mar 1;10(3):799-815. eCollection 2020. Am J Cancer Res. 2020. PMID: 32266092 Free PMC article.
-
Combination Cancer Therapy Using Chimeric Antigen Receptor-Engineered Natural Killer Cells as Drug Carriers.Mol Ther. 2017 Dec 6;25(12):2607-2619. doi: 10.1016/j.ymthe.2017.08.010. Epub 2017 Aug 19. Mol Ther. 2017. PMID: 28919377 Free PMC article.
-
Wogonoside inhibits cell growth and induces mitochondrial-mediated autophagy-related apoptosis in human colon cancer cells through the PI3K/AKT/mTOR/p70S6K signaling pathway.Oncol Lett. 2018 Apr;15(4):4463-4470. doi: 10.3892/ol.2018.7852. Epub 2018 Jan 24. Oncol Lett. 2018. PMID: 29541215 Free PMC article.
-
Optimization of Formulations Consisting of Layered Double Hydroxide Nanoparticles and Small Interfering RNA for Efficient Knockdown of the Target Gene.ACS Omega. 2018 May 31;3(5):4871-4877. doi: 10.1021/acsomega.8b00397. Epub 2018 May 2. ACS Omega. 2018. PMID: 30023905 Free PMC article.
References
-
- Davies JM, Goldberg RM. Treatment of metastatic colorectal cancer. Semin Oncol. 2011;38(4):552–560. - PubMed
-
- Saville M, Lietzau J, Pluda J, et al. Treatment of HIV-associated Kaposi’s sarcoma with paclitaxel. Lancet. 1995;346(8966):26–28. - PubMed
-
- Moktan S, Ryppa C, Kratz F, Raucher D. A thermally responsive biopolymer conjugated to an acid-sensitive derivative of paclitaxel stabilizes microtubules, arrests cell cycle, and induces apoptosis. Invest New Drugs. 2012;30(1):236–248. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

