Membrane Insertion for the Detection of Lipopolysaccharides: Exploring the Dynamics of Amphiphile-in-Lipid Assays
- PMID: 27227979
- PMCID: PMC4881986
- DOI: 10.1371/journal.pone.0156295
Membrane Insertion for the Detection of Lipopolysaccharides: Exploring the Dynamics of Amphiphile-in-Lipid Assays
Abstract
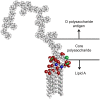
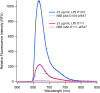
Shiga toxin-producing Escherichia coli is an important cause of foodborne illness, with cases attributable to beef, fresh produce and other sources. Many serotypes of the pathogen cause disease, and differentiating one serotype from another requires specific identification of the O antigen located on the lipopolysaccharide (LPS) molecule. The amphiphilic structure of LPS poses a challenge when using classical detection methods, which do not take into account its lipoglycan biochemistry. Typically, detection of LPS requires heat or chemical treatment of samples and relies on bioactivity assays for the conserved lipid A portion of the molecule. Our goal was to develop assays to facilitate the direct and discriminative detection of the entire LPS molecule and its O antigen in complex matrices using minimal sample processing. To perform serogroup identification of LPS, we used a method called membrane insertion on a waveguide biosensor, and tested three serogroups of LPS. The membrane insertion technique allows for the hydrophobic association of LPS with a lipid bilayer, where the exposed O antigen can be targeted for specific detection. Samples of beef lysate were spiked with LPS to perform O antigen specific detection of LPS from E. coli O157. To validate assay performance, we evaluated the biophysical interactions of LPS with lipid bilayers both in- and outside of a flow cell using fluorescence microscopy and fluorescently doped lipids. Our results indicate that membrane insertion allows for the qualitative and reliable identification of amphiphilic LPS in complex samples like beef homogenates. We also demonstrated that LPS-induced hole formation does not occur under the conditions of the membrane insertion assays. Together, these findings describe for the first time the serogroup-specific detection of amphiphilic LPS in complex samples using a membrane insertion assay, and highlight the importance of LPS molecular conformations in detection architectures.
Conflict of interest statement
Figures




References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

