Peroxo and Superoxo Moieties Bound to Copper Ion: Electron-Transfer Equilibrium with a Small Reorganization Energy
- PMID: 27228314
- PMCID: PMC4950875
- DOI: 10.1021/jacs.6b02404
Peroxo and Superoxo Moieties Bound to Copper Ion: Electron-Transfer Equilibrium with a Small Reorganization Energy
Abstract
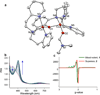
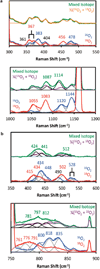
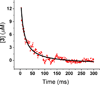
Oxygenation of [Cu2(UN-O(-))(DMF)](2+) (1), a structurally characterized dicopper Robin-Day class I mixed-valent Cu(II)Cu(I) complex, with UN-O(-) as a binucleating ligand and where dimethylformamide (DMF) binds to the Cu(II) ion, leads to a superoxo-dicopper(II) species [Cu(II)2(UN-O(-))(O2(•-))](2+) (2). The formation kinetics provide that kon = 9 × 10(-2) M(-1) s(-1) (-80 °C), ΔH(‡) = 31.1 kJ mol(-1) and ΔS(‡) = -99.4 J K(-1) mol(-1) (from -60 to -90 °C data). Complex 2 can be reversibly reduced to the peroxide species [Cu(II)2(UN-O(-))(O2(2-))](+) (3), using varying outer-sphere ferrocene or ferrocenium redox reagents. A Nernstian analysis could be performed by utilizing a monodiphenylamine substituted ferrocenium salt to oxidize 3, leading to an equilibrium mixture with Ket = 5.3 (-80 °C); a standard reduction potential for the superoxo-peroxo pair is calculated to be E° = +130 mV vs SCE. A literature survey shows that this value falls into the range of biologically relevant redox reagents, e.g., cytochrome c and an organic solvent solubilized ascorbate anion. Using mixed-isotope resonance Raman (rRaman) spectroscopic characterization, accompanied by DFT calculations, it is shown that the superoxo complex consists of a mixture of μ-1,2- (2(1,2)) and μ-1,1- (2(1,1)) isomers, which are in rapid equilibrium. The electron transfer process involves only the μ-1,2-superoxo complex [Cu(II)2(UN-O(-))(μ-1,2-O2(•-))](2+) (2(1,2)) and μ-1,2-peroxo structures [Cu(II)2(UN-O(-))(O2(2-))](+) (3) having a small bond reorganization energy of 0.4 eV (λin). A stopped-flow kinetic study results reveal an outer-sphere electron transfer process with a total reorganization energy (λ) of 1.1 eV between 2(1,2) and 3 calculated in the context of Marcus theory.
Figures

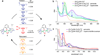

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

