Toxicity Assessment of a Phase III Study Evaluating FEC-Doc and FEC-Doc Combined with Gemcitabine as an Adjuvant Treatment for High-Risk Early Breast Cancer: the SUCCESS-A Trial
- PMID: 27239063
- PMCID: PMC4873296
- DOI: 10.1055/s-0042-106209
Toxicity Assessment of a Phase III Study Evaluating FEC-Doc and FEC-Doc Combined with Gemcitabine as an Adjuvant Treatment for High-Risk Early Breast Cancer: the SUCCESS-A Trial
Abstract
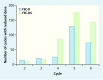
Introduction: This paper aims to evaluate the toxicity profile of additive gemcitabine to adjuvant taxane-based chemotherapy in breast cancer patients. Methods: Patients enrolled in this open-label randomized controlled Phase III study were treated with 3 cycles of epirubicin-fluorouracil-cyclophosphamide (FEC) chemotherapy followed by 3 cycles of docetaxel with those receiving 3 cycles of FEC followed by 3 cycles of gemcitabine-docetaxel (FEC-DG). 3690 patients were evaluated according to National Cancer Institute (NCI) toxicity criteria (CTCAE). The study medications were assessed by the occurrence of grade 3-4 adverse events, dose reductions, postponements of treatment cycles and granulocyte colony-stimulating factor (G-CSF) support. Results: No differences in neutropenia or febrile neutropenia were demonstrated. However, thrombocytopenia was significantly increased with FEC-DG treatment (2.0 vs. 0.5 %, p < 0.001), as was leukopenia (64.1 vs. 58.5 %, p < 0.001). With FEC-DG significantly more G-CSF support in cycles 4 to 6 (FEC-DG: 57.8 %, FEC-D: 36.3 %, p < 0.001) was provided. Transaminase elevation was significantly more common with FEC-DG (SGPT: 6.3 %, SGOT: 2 %), whereas neuropathy (1.2 %), arthralgia (1.6 %) and bone pain (2.6 %) were more common using FEC-D. Dose reductions > 20 % (4 vs. 2.4 %) and postponement of treatment cycles (0.9 vs. 0.4 %) were significantly more frequent in the FEC-DG arm. Eight deaths occurred during treatment in the FEC-DG arm and four in the FEC-D arm. Conclusion: The addition of gemcitabine increased hematological toxicity and was associated with more dose reductions and postponements of treatment cycles.
Einleitung: Die vorliegende Studie untersucht das Toxizitätsprofil nach der zusätzlichen Gabe von Gemcitabin in Kombination mit einer adjuvanten Taxan-basierten Chemotherapie bei Patientinnen mit Brustkrebs. Methode: Es handelt sich hier um eine randomisierte kontrollierte Phase-III-Open-Label-Studie. Alle in der Studie aufgenommenen Patientinnen erhielten 3 Zyklen Epirubicin-Fluorouracil-Cyclophosphamid (FEC) gefolgt von 3 Zyklen Docetaxel bzw. 3 Zyklen FEC gefolgt von 3 Zyklen Gemcitabin-Docetaxel (FEC-DG). Die Daten von insgesamt 3690 Patientinnen wurden mittels der Toxizitätskriterien (CTCAE) des nationalen Krebsinstituts der USA (National Cancer Institute [NCI]) ausgewertet. Kriterien für die Auswertung der Studienmedikation waren das Auftreten unerwünschter Ereignisse 3. oder 4. Grades, Dosisreduktionen, Verschiebungen nachfolgender Behandlungszyklen und Granulozytenkolonie-stimulierender Faktor (G-CSF)-Gabe. Ergebnisse: Es zeigten sich keine Unterschiede zwischen den beiden Gruppen hinsichtlich der Entwicklung einer Neutropenie oder einer febrilen Neutropenie. Dagegen war die Thrombozytopenie nach der Behandlung mit FEC-DG signifikant erhöht (2,0 vs. 0,5 %, p < 0,001), und die Leukopenie trat in dieser Gruppe ebenfalls deutlich häufiger auf (64,1 vs. 58,5 %, p < 0,001). Der Einsatz von G-CSF-Präparaten in den Zyklen 4–6 war deutlich höher in der FEC-DG-Gruppe (FEC-DG: 57,8 %, FEC-D: 36,3 %, p < 0,001). Ein Transaminasenanstieg kam wesentlich häufiger in der FEC-DG-Gruppe vor (SGPT: 6,3 %, SGOT: 2 %), während Neuropathien (1,2 %), Arthralgien (1,6 %) und Knochenschmerzen (2,6 %) häufiger in der FEC-D-Gruppe auftraten. Dosisreduktionen > 20 % (4 vs. 2.4 %) sowie eine Verschiebung nachfolgender Behandlungszyklen (0,9 vs. 0,4 %) kamen deutlich häufiger im FEC-DG-Studienarm vor. Während der Behandlung starben 8 Patientinnen im FEC-DG-Studienarm und 4 im FEC-D-Studienarm. Schlussfolgerung: Die zusätzliche Gabe von Gemcitabin hat die hämatologische Toxizität erhöht und war mit mehr Dosisreduktionen und mehr Verschiebungen nachfolgender Behandlungszyklen assoziiert.
Keywords: adjuvant chemotherapy; breast cancer; gemcitabine; hematological; toxicity.
Conflict of interest statement
Figures
References
-
- Early Breast Cancer Trialistʼs Collaborative Group (EBCTCG) . Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;365:1687–1717. - PubMed
-
- Petrelli F, Borgonovo K, Cabiddu M. et al. Mortality, leukemic risk, and cardiovascular toxicity of adjuvant anthracycline and taxane chemotherapy in breast cancer: a meta-analysis. Breast Cancer Res Treat. 2012;135:335–346. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources