Jasmonic Acid Modulates the Physio-Biochemical Attributes, Antioxidant Enzyme Activity, and Gene Expression in Glycine max under Nickel Toxicity
- PMID: 27242811
- PMCID: PMC4864666
- DOI: 10.3389/fpls.2016.00591
Jasmonic Acid Modulates the Physio-Biochemical Attributes, Antioxidant Enzyme Activity, and Gene Expression in Glycine max under Nickel Toxicity
Abstract
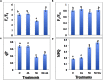
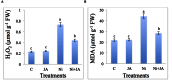
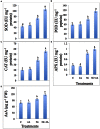
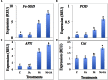
In present study, we evaluated the effects of Jasmonic acid (JA) on physio-biochemical attributes, antioxidant enzyme activity, and gene expression in soybean (Glycine max L.) plants subjected to nickel (Ni) stress. Ni stress decreases the shoot and root length and chlorophyll content by 37.23, 38.31, and 39.21%, respectively, over the control. However, application of JA was found to improve the chlorophyll content and length of shoot and root of Ni-fed seedlings. Plants supplemented with JA restores the chlorophyll fluorescence, which was disturbed by Ni stress. The present study demonstrated increase in proline, glycinebetaine, total protein, and total soluble sugar (TSS) by 33.09, 51.26, 22.58, and 49.15%, respectively, under Ni toxicity over the control. Addition of JA to Ni stressed plants further enhanced the above parameters. Ni stress increases hydrogen peroxide (H2O2) by 68.49%, lipid peroxidation (MDA) by 50.57% and NADPH oxidase by 50.92% over the control. Supplementation of JA minimizes the accumulation of H2O2, MDA, and NADPH oxidase, which helps in stabilization of biomolecules. The activities of superoxide dismutase (SOD), peroxidase (POD), catalase (CAT), and ascorbate peroxidase (APX) increases by 40.04, 28.22, 48.53, and 56.79%, respectively, over the control in Ni treated seedlings and further enhancement in the antioxidant activity was observed by the application of JA. Ni treated soybean seedlings showed increase in expression of Fe-SOD by 77.62, CAT by 15.25, POD by 58.33, and APX by 80.58% over the control. Nevertheless, application of JA further enhanced the expression of the above genes in the present study. Our results signified that Ni stress caused negative impacts on soybean seedlings, but, co-application of JA facilitate the seedlings to combat the detrimental effects of Ni through enhanced osmolytes, activity of antioxidant enzymes and gene expression.
Keywords: antioxidants; growth; jasmonic acid; lipid peroxidation; nickel stress; osmolytes; reactive oxygen species; soybean.
Figures
References
-
- Abdel Latef A. A. M., Jan S., Abd Allah E. F., Rashid B., John R., Ahmad P. (2016). “Soybean under abiotic stress: proteomic approach,” in Plant-Environment Interaction: Responses and Approaches to Mitigate Stress, eds Azooz M. M., Ahmad P. (Chichester: John Wiley; ), 28–42.
-
- Abu-Romman S., Shatnawi M. (2011). Isolation and expression analysis of chloroplastic copper/zinc superoxide dismutase gene in barley. S. Afr. J. Bot. 77 328–334. 10.1016/j.sajb.2010.09.012 - DOI
-
- Ahmad P. (2013). Oxidative Damage to Plants, Antioxidant Networks and Signaling. Cambridge, MA: Academic Press.
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

