Signal transduction pathways activated by insulin-like peptide 5 at the relaxin family peptide RXFP4 receptor
- PMID: 27243554
- PMCID: PMC5406383
- DOI: 10.1111/bph.13522
Signal transduction pathways activated by insulin-like peptide 5 at the relaxin family peptide RXFP4 receptor
Erratum in
-
Correction.Br J Pharmacol. 2017 Dec;174(24):4836. doi: 10.1111/bph.14111. Br J Pharmacol. 2017. PMID: 29235105 Free PMC article. No abstract available.
Abstract
Background and purpose: Insulin-like peptide 5 (INSL5) is a two-chain, three-disulfide-bonded peptide of the insulin/relaxin superfamily, uniquely expressed in enteroendocrine L-cells of the colon. It is the cognate ligand of relaxin family peptide RXFP4 receptor that is mainly expressed in the colorectum and enteric nervous system. This study identifies new signalling pathways activated by INSL5 acting on RXFP4 receptors.
Experimental approach: INSL5/RXFP4 receptor signalling was investigated using AlphaScreen® proximity assays. Recruitment of Gαi/o proteins by RXFP4 receptors was determined by rescue of Pertussis toxin (PTX)-inhibited cAMP and ERK1/2 responses following transient transfection of PTX-insensitive Gαi/o C351I mutants. Cell proliferation was studied with bromodeoxyuridine. RXFP4 receptor interactions with β-arrestins, GPCR kinase 2 (GRK2), KRas and Rab5a was assessed with real-time BRET. Gene expression was investigated using real-time quantitative PCR. Insulin release was measured using HTRF and intracellular Ca2+ flux monitored in a Flexstation® using Fluo-4-AM.
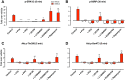
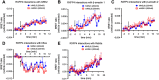
Key results: INSL5 inhibited forskolin-stimulated cAMP accumulation and increased phosphorylation of ERK1/2, p38MAPK, Akt Ser473 , Akt Thr308 and S6 ribosomal protein. cAMP and ERK1/2 responses were abolished by PTX and rescued by mGαoA , mGαoB and mGαi2 and to a lesser extent mGαi1 and mGαi3 . RXFP4 receptors interacted with GRK2 and β-arrestins, moved towards Rab5a and away from KRas, indicating internalisation following receptor activation. INSL5 inhibited glucose-stimulated insulin secretion and Ca2+ mobilisation in MIN6 insulinoma cells and forskolin-stimulated cAMP accumulation in NCI-H716 enteroendocrine cells.
Conclusions and implications: Knowledge of signalling pathways activated by INSL5 at RXFP4 receptors is essential for understanding the biological roles of this novel gut hormone.
Linked articles: This article is part of a themed section on Recent Progress in the Understanding of Relaxin Family Peptides and their Receptors. To view the other articles in this section visit http://onlinelibrary.wiley.com/doi/10.1111/bph.v174.10/issuetoc.
© 2016 The British Pharmacological Society.
Figures





References
-
- Bahia DS, Wise A, Fanelli F, Lee M, Rees S, Milligan G (1998). Hydrophobicity of residue351 of the G protein Gi1α alpha determines the extent of activation by the α2A‐adrenoceptor. Biochemistry 37: 11555–11562. - PubMed
-
- Belgi A, Bathgate RA, Kocan M, Patil N, Zhang S, Tregear GW et al. (2013). Minimum active structure of insulin‐like peptide 5. J Med Chem 56: 9509–9516. - PubMed
-
- Belgi A, Hossain MA, Shabanpoor F, Chan L, Zhang S, Bathgate RA et al. (2011). Structure and function relationship of murine insulin‐like peptide 5 (INSL5): free C‐terminus is essential for RXFP4 receptor binding and activation. Biochemistry 50: 8352–8361. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

